Advertisements
Advertisements
Write balanced equation for the reaction between iron and dilute sulphuric acid.
Concept: undefined >> undefined
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Copper carbonate
Concept: undefined >> undefined
Advertisements
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Lead nitrate solution
Concept: undefined >> undefined
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Zinc hydroxide
Concept: undefined >> undefined
In this question, you required to supply the word (or words) that will make the sentence correct. Rewrite the copper statement.
Copper sulphate crystals are dehydrate by sulphuric acid.
Concept: undefined >> undefined
What do you see when concentrated sulphuric acid is added to copper sulphate-5-water ( You are not required to say what is happing, nor is it necessary to name the products.)
Concept: undefined >> undefined
Copy and complete the following table.
Column 3 has the names of gases to be prepared using the substance you enter in column 1, along with dilute or concentrated sulphuric acid, as indicated in column 2.
| Column 1 | Column 2 | Column 3 |
| Substance reacted with acid | Dilute or concentrated sulphuric acid | Gas |
| Hydrogen | ||
| Carbon dioxide | ||
| Only chlorine |
Write the equation for the laboratory preparation of :
(i) Sodium sulphate using dilute sulphuric acid.
(ii) Lead sulphate using dilute sulphuric acid.
Concept: undefined >> undefined
Which concentrated acid oxidizes sulphur directly to sulphuric acid ? Write the equation for the same. What is the name of the process by which sulphuric acid is manufactured ? Name the catalyst used in the process.
Concept: undefined >> undefined
Complete the following sentence, choosing the correct word from the given options.
"Concentrated sulphuric acid is used in the laboratory preparation of nitric acid and hydrochloric acid because it is ____________ in comparison to these two acids."
Concept: undefined >> undefined
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Iron (II) sulphate from iron.
Concept: undefined >> undefined
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Copper sulphate from copper.
Concept: undefined >> undefined
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Lead sulphate from lead nitrate.
Concept: undefined >> undefined
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Sodium Sulphate from sodium carbonate.
Concept: undefined >> undefined
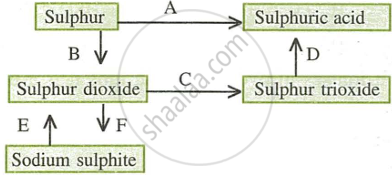
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Concept: undefined >> undefined
Write balanced equation for potassium hydrogen carbonate and dilute sulphuric acid.
Concept: undefined >> undefined
A, B, C and D summarize the properties of sulphuric acid depending on whether it is dilute or concentrated. Choose the property (A, B, C or D), depending on which is relevant to each of the preparations (i) to (ii).
A. Dilute acid (typical acid properties)
B. Non-volatile acid
C. Oxidizing agent
D. Dehydrating agent
(i) Preparation of hydrogen chloride.
(ii) Preparation of ethane from ethanol
(iii) Preparation of copper sulphate from copper oxide.
Concept: undefined >> undefined
Name the gas evolved in following case:
The gas produced by the action of concentrated sulphuric acid on sodium chloride.
Concept: undefined >> undefined
Write the equation of the following reaction:
Sulphur is heated with concentrated sulphuric acid.
Concept: undefined >> undefined
Write the equation of the following reaction :
Concentrated sulphuric acid is poured over sugar
Concept: undefined >> undefined
State your observation for the following case :
Moist blue litmus is introduced into a gas jar of sulphur dioxide.
Concept: undefined >> undefined
