Advertisements
Advertisements
Which of the following transition metals shows +1 and +2 oxidation states?
Concept: undefined >> undefined
Assertion (A): Transition metals show their highest oxidation state with oxygen.
Reason (R): The ability of oxygen to form multiple bonds to metals.
Concept: undefined >> undefined
Advertisements
Do the following conversions in not more than two steps:
CH3COOH to CH3COCH3
Concept: undefined >> undefined
Do the following conversions in not more than two steps:
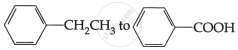
Concept: undefined >> undefined
Which property of transition metals enables them to behave as catalysts?
Concept: undefined >> undefined
Why is the C-O bond length in phenols less than that in methanol?
Concept: undefined >> undefined
Complete the following reaction and justify that it is a disproportionation reaction:
\[\ce{3MnO^{2-}4 + 4H^+ -> \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{} + 2H2O}\]
Concept: undefined >> undefined
The given graph shows the trends in melting points of transition metals:

Explain the reason why Cr has the highest melting point and manganese (Mn) has a lower melting point.
Concept: undefined >> undefined
Consider the following standard electrode potential values:
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\], E0 = +0.77 V
\[\ce{MnO^{-4}_{ (aq)} + 8H^+ + 5e^- -> Mn^{2+}_{ (aq)} + 4H2O_{(l)}}\], E0 = +1.51 V
What is the cell potential for the redox reaction?
Concept: undefined >> undefined
Which of the following ions has the electronic configuration 3d6?
(Atomic number: Mn = 25, Co = 27, Ni = 28)
Concept: undefined >> undefined
Which of the following statements is not true about glucose?
Concept: undefined >> undefined
A transition element X has an electronic configuration [Ar]4s23d3. Predict its likely oxidation states.
Concept: undefined >> undefined
Consider the following standard electrode potential values:
\[\ce{Sn^{2+}_{ (aq)} + 2e^- -> Sn_{(s)}}\]; E0 = −0.14 V
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\]; E0 = +0.77 V
What is the cell reaction and potential for the spontaneous reaction that occurs?
Concept: undefined >> undefined
Write the number of unpaired electrons in Cr3+.
(Atomic number of Cr = 24)
Concept: undefined >> undefined
Account for the following:
Copper has an exceptionally positive `"E"_("M"^(2+)//"M")^0` value.
Concept: undefined >> undefined
Account for the following:
Eu2+ with electronic configuration [Xe]4f76s2 is a strong reducing agent.
Concept: undefined >> undefined
The trend of which property is represented by the following graph?

Concept: undefined >> undefined
Radioactive decay follows first-order kinetics. The initial amount of two radioactive elements X and Y is 1 gm each. What will be the ratio of X and Y after two days if their half-lives are 12 hours and 16 hours respectively?
Concept: undefined >> undefined
In order to protect iron from corrosion, which one will you prefer as a sacrificial electrode, Ni or Zn? Why? (Given standard electrode potentials of Ni, Fe and Zn are -0.25 V, -0.44 V and -0.76 V respectively.)
Concept: undefined >> undefined
The second ionization enthalpies of chromium and manganese are 1592 and 1509 kJ/mol respectively. Explain the lower value of Mn.
Concept: undefined >> undefined
