Advertisements
Advertisements
Observe the graph shown in figure and answer the following questions:
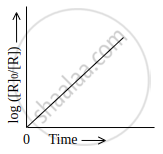
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
Concept: undefined >> undefined
Write three differences between physisorption and chemisorption.
Concept: undefined >> undefined
Advertisements
Give reasons for the following statement:
Transition metals and most of their compounds show paramagnetic behaviour.
Concept: undefined >> undefined
Give reasons for the following statement:
Zn, Cd, and Hg are soft metals.
Concept: undefined >> undefined
Give reason for the following statement:
[Ti(H2O)]3+ is coloured while [Sc(H2O)6]3+ is colourless.
Concept: undefined >> undefined
Give reason for the following statement:
Physical and chemical properties of the 4d and 5d series of the transition elements are quite similar to expected.
Concept: undefined >> undefined
An organic compound (A) with molecular formula C3H7NO on heating with Br2 and KOH forms a compound (B). Compound (B) on heating with CHCl3 and alcoholic KOH produces a foul-smelling compound (C) and on reacting with C6H5SO2Cl forms a compound (D) which is soluble in alkali. Write the structure of (A), (B), (C) and (D).
Concept: undefined >> undefined
Account for the following:
Transition metals form alloys.
Concept: undefined >> undefined
Why Zn, Cd and Hg are not called transition metals?
Concept: undefined >> undefined
How is the variability in oxidation states of transition metals different from that of p-block elements?
Concept: undefined >> undefined
The oxidation state of Fe in [Fe(CO)5] is ______.
Concept: undefined >> undefined
The slope in the plot of ln[R] vs. time for a first order reaction is ______.
Concept: undefined >> undefined
Which of the following characteristics of transition metals is associated with their catalytic activity?
Concept: undefined >> undefined
Do the following conversions in not more than two steps:
\[\begin{array}{cc}
\ce{CH3CN to CH3 - C - CH3}\\
\phantom{...........}||\\
\phantom{...........}\ce{O}
\end{array}\]
Concept: undefined >> undefined
Do the following conversions in not more than two steps:

Concept: undefined >> undefined
Account for the following:
Sc3+ is colourless whereas Ti3+ is coloured in an aqueous solution.
Concept: undefined >> undefined
Account for the following:
Ce4+ is a strong oxidising agent.
Concept: undefined >> undefined
Assertion (A): Transition metals have high enthalpy of atomisation.
Reason (R): Greater number of unpaired electrons in transition metals results in weak metallic bonding.
Concept: undefined >> undefined
Which one among the following metals of the 3d series has the lowest melting point?
Concept: undefined >> undefined
The slope in the plot of `log ["R"]_0/(["R"])` Vs. time for a first-order reaction is ______.
Concept: undefined >> undefined
