Advertisements
Advertisements
How do you explain the amphoteric behaviour of amino acids?
Concept: undefined >> undefined
Draw the structure of the given derivative.
Acetaldehydedimethylacetal
Concept: undefined >> undefined
Advertisements
Write the reactions of Williamson synthesis of 2-ethoxy-3-methylpentane starting from ethanol and 3-methylpentan-2-ol.
Concept: undefined >> undefined
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
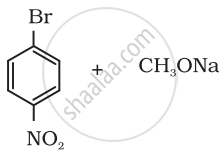 |
 |
| (i) | (ii) |
Concept: undefined >> undefined
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
1-Propoxypropane
Concept: undefined >> undefined
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
Ethoxybenzene
Concept: undefined >> undefined
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Concept: undefined >> undefined
Write the name of the reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
1-Methoxyethane
Concept: undefined >> undefined
Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
Concept: undefined >> undefined
How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of this reaction.
Concept: undefined >> undefined
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
Concept: undefined >> undefined
Explain the following with an example.
Williamson ether synthesis
Concept: undefined >> undefined
How will you bring about the following conversion?
Benzene to biphenyl
Concept: undefined >> undefined
How the following conversion can be carried out?
Benzene to diphenyl
Concept: undefined >> undefined
Explain effects of lanthanoid contraction.
Concept: undefined >> undefined
What are the consequences of lanthanoid contraction?
Concept: undefined >> undefined
Write the structure of 3-Bromo-2-methylprop-1-ene
Concept: undefined >> undefined
Write the formula of the compound of iodine which is obtained when conc. HNO3 oxidises I2
Concept: undefined >> undefined
Write one difference in Multimolecular colloid and Associated colloid
Concept: undefined >> undefined
Give reasons for the following: N2O5 is more acidic than N2O3
Concept: undefined >> undefined
