Please select a subject first
Advertisements
Advertisements
Following are the transition metal ions of 3d series:
Ti4+, V2+, Mn3+, Cr3+
(Atomic numbers: Ti = 22, V = 23, Mn = 25, Cr = 24)
Answer the following:
1) Which ion is most stable in an aqueous solution and why?
2) Which ion is a strong oxidising agent and why?
3) Which ion is colourless and why?
Concept: undefined >> undefined
How would you account for the following?
Zr (Z = 40) and Hf (Z = 72) have almost identical radii.
Concept: undefined >> undefined
Advertisements
How would you account for the following?
Transition metals and their compounds act as catalysts.
Concept: undefined >> undefined
An analysis shows that FeO has a non-stoichiometric composition with formula Fe0.95O. Give reason.
Concept: undefined >> undefined
Complete and balance the following chemical equations
`Fe^(2+) + MnO_4^(-) + H^+ ->`
Concept: undefined >> undefined
Complete and balance the following chemical equations
`MnO_4^(-) + H_2O + I^(-) ->`
Concept: undefined >> undefined
Give reasons:
E° value for the Mn3+/Mn2+ couple is much more positive than that for Fe3+/Fe2+.
Concept: undefined >> undefined
Give reasons Iron has the higher enthalpy of atomization than that of copper.
Concept: undefined >> undefined
Give reasons: Sc3+ is colourless in aqueous solution whereas Ti3+ is coloured.
Concept: undefined >> undefined
Shyam went to a grocery shop to purchase some food items. The shopkeeper packed all the items in polythene bags and gave them to Shyam. But Shyam refused to accept the polythene bags and asked the shopkeeper to pack the items in paper bags. He informed the shopkeeper about the heavy penalty imposed by the government for using polythene bags. The shopkeeper promised that he would use paper bags in future in place of polythene bags.
1) Write the values (at least two) shown by Shyam.
2) Write one structural difference between low-density polyethene and high-density polyethene.
3) Why did Shyam refuse to accept the items in polythene bags?
4) What is a biodegradable polymer? Give an example.
Concept: undefined >> undefined
Write the cell reaction and calculate the e.m.f of the following cell at 298 K:
Sn(s) | Sn2+ (0.004 M) || H+ (0.020 M) | H2(g) (1 bar) | Pt(s)
(Given: `E_(Sn^(2+)"/"Sn)^° = -0.14` V)
Concept: undefined >> undefined
Write the IUPAC name of the given compound :
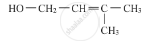
Concept: undefined >> undefined
Physisorption is reversible while chemisorption is irreversible. Why ?
Concept: undefined >> undefined
Why do transition elements show variable oxidation states ? In 3d series (Sc to Zn), which elements shows the maximum number of oxidation state and why ?
Concept: undefined >> undefined
Give reasons for the following observations :
Powdered substances are more effective adsorbents.
Concept: undefined >> undefined
Account for the following :
Bond angle in NH+4NH4+ is higher than NH3.
Concept: undefined >> undefined
Give simple chemical tests to distinguish between the following pair of compounds:
Benzaldehyde and Benzoic acid
Concept: undefined >> undefined
Which component of starch is a branched polymer of α-glucose and insoluble in water?
Concept: undefined >> undefined
Using IUPAC norms, write the formula of Hexaamminecobalt(III) sulphate.
Concept: undefined >> undefined
Tendency to show –3 oxidation state decrease from Nitrogn (N) to Bismuth (Bi).
Concept: undefined >> undefined
