Advertisements
Advertisements
Find the ratio of the lengths of an iron rod and an aluminium rod for which the difference in the lengths is independent of temperature. Coefficients of linear expansion of iron and aluminium are 12 × 10–6 °C–1 and 23 × 10–6 °C–1 respectively.
Concept: undefined >> undefined
An aluminium plate fixed in a horizontal position has a hole of diameter 2.000 cm. A steel sphere of diameter 2.005 cm rests on this hole. All the lengths refer to a temperature of 10 °C. The temperature of the entire system is slowly increased. At what temperature will the ball fall down? Coefficient of linear expansion of aluminium is 23 × 10–6 °C–1 and that of steel is 11 × 10–6 °C–1.
Concept: undefined >> undefined
Advertisements
A glass window is to be fit in an aluminium frame. The temperature on the working day is 40°C and the glass window measures exactly 20 cm × 30 cm. What should be the size of the aluminium frame so that there is no stress on the glass in winter even if the temperature drops to 0°C? Coefficients of linear expansion for glass and aluminium are 9.0 × 10–6 °C–1 and 24 ×100–6°C–1 , respectively.
Concept: undefined >> undefined
Does a gas have just two specific heat capacities or more than two? Is the number of specific heat capacities of a gas countable?
Concept: undefined >> undefined
Can we define specific heat capacity at constant temperature?
Concept: undefined >> undefined
Can we define specific heat capacity for an adiabatic process?
Concept: undefined >> undefined
Does a solid also have two kinds of molar heat capacities Cp and Cv? If yes, is Cp > Cv? Or is Cp − Cv = R?
Concept: undefined >> undefined
In a real gas, the internal energy depends on temperature and also on volume. The energy increases when the gas expands isothermally. Examining the derivation of Cp − Cv = R, find whether Cp − Cv will be more than R, less than R or equal to R for a real gas.
Concept: undefined >> undefined
Can a process on an ideal gas be both adiabatic and isothermal?
Concept: undefined >> undefined
Show that the slope of the p−V diagram is greater for an adiabatic process compared to an isothermal process.
Concept: undefined >> undefined
Can two states of an ideal gas be connected by an isothermal process as well as an adiabatic process?
Concept: undefined >> undefined
In a room containing air, heat can go from one place to another
Concept: undefined >> undefined
In an isothermal process on an ideal gas, the pressure increases by 0.5%. The volume decreases by about
Concept: undefined >> undefined
Two samples A and B are initially kept in the same state. Sample A is expanded through an adiabatic process and the sample B through an isothermal process. The final volumes of the samples are the same. The final pressures in A and B are pA and pBrespectively.
Concept: undefined >> undefined
Let ∆Wa and ∆Wb be the work done by the systems A and B, respectively, in the previous question.
Concept: undefined >> undefined
Consider the processes A and B shown in the figure. It is possible that
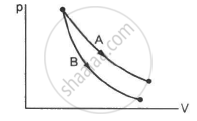
Concept: undefined >> undefined
Three identical adiabatic containers A, B and C contain helium, neon and oxygen, respectively, at equal pressure. The gases are pushed to half their original volumes.
(a) The final temperatures in the three containers will be the same.
(b) The final pressures in the three containers will be the same.
(c) The pressures of helium and neon will be the same but that of oxygen will be different.
(d) The temperatures of helium and neon will be the same but that of oxygen will be different.
Concept: undefined >> undefined
5 g of a gas is contained in a rigid container and is heated from 15°C to 25°C. Specific heat capacity of the gas at constant volume is 0.172 cal g−1 °C−1 and the mechanical equivalent of heat is 4.2 J cal−1. Calculate the change in the internal energy of the gas
Concept: undefined >> undefined
A sample of air weighing 1.18 g occupies 1.0 × 103 cm3 when kept at 300 K and 1.0 × 105 Pa. When 2.0 cal of heat is added to it at constant volume, its temperature increases by 1°C. Calculate the amount of heat needed to increase the temperature of air by 1°C at constant pressure if the mechanical equivalent of heat is 4.2 × 107 erg cal−1. Assume that air behaves as an ideal gas.
Concept: undefined >> undefined
An ideal gas expands from 100 cm3 to 200 cm3 at a constant pressure of 2.0 × 105 Pa when 50 J of heat is supplied to it. Calculate (a) the change in internal energy of the gas (b) the number of moles in the gas if the initial temperature is 300 K (c) the molar heat capacity Cp at constant pressure and (d) the molar heat capacity Cv at constant volume.
Concept: undefined >> undefined
