Please select a subject first
Advertisements
Advertisements
Which of the following ions will cause hardness in water sample?
Concept: undefined >> undefined
Which of the following compounds is used for water softening?
Concept: undefined >> undefined
Advertisements
Hardness of water may be temporary or permanent. Permanent hardness is due to the presence of:
(i) Chlorides of \[\ce{Ca}\] and \[\ce{Mg}\] in water.
(ii) Sulphates of \[\ce{Ca}\] and \[\ce{Mg}\] in water.
(iii) Hydrogen carbonates of \[\ce{Ca}\] and \[\ce{Mg}\] in water.
(iv) Carbonates of alkali metals in water.
Concept: undefined >> undefined
Complete the following equations:
\[\ce{PbS (s) + H2O2 (aq) ->}\]
Concept: undefined >> undefined
Complete the following equations:
\[\ce{CO (g) + 2H2 (g) ->[Cobalt][Catalyst]}\]
Concept: undefined >> undefined
Discuss briefly de-mineralisation of water by ion exchange resin.
Concept: undefined >> undefined
Why is the Ionisation enthalpy of hydrogen higher than that of sodium?
Concept: undefined >> undefined
Match the items in Column I with the relevant item in Column II.
| Column I | Column II |
| (i) Hydrogen peroxide is used as a | (a) zeolite |
| (ii) Used in Calgon method | (b) perhydrol |
| (iii) Permanent hardness of hard water is removed by | (c) sodium hexametaphosphate |
| (d) propellant |
Concept: undefined >> undefined
Assertion (A): Permanent hardness of water is removed by treatment with washing soda.
Reason (R): Washing soda reacts with soluble magnesium and calcium sulphate to form insoluble carbonates.
Concept: undefined >> undefined
If a trivalent atom replaces a few silicon atoms in three-dimensional network of silicon dioxide, what would be the type of charge on overall structure?
Concept: undefined >> undefined
Match the ions given in Column I with their nature given in Column II.
| Column I | Column II |
| (i) \[\ce{CH3 - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - \overset{⊕}{C}H - CH3}\] | (a) Stable due to resonance |
| (ii) \[\ce{F3 - \overset{⊕}{C}}\] | (b) Destabilised due to inductive effect |
|
(iii) \[\begin{array}{cc} |
(c) Stabilised by hyperconjugation |
| (iv) \[\ce{CH3 - \overset{⊕}{C}H - CH3}\] | (d) A secondary carbocation |
Concept: undefined >> undefined
Arrange the halogens F2, Cl2, Br2, I2, in order of their increasing reactivity with alkanes.
Concept: undefined >> undefined
The addition of HBr to 1-butene gives a mixture of products A, B and C
| (A) | 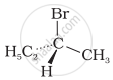 |
| (B) |  |
| (C) | CH3 – CH2 – CH2 – CH2 – Br |
The mixture consists of:
Concept: undefined >> undefined
Arrange the following hydrogen halides in order of their decreasing reactivity with propene.
Concept: undefined >> undefined
Which of the following alkenes on ozonolysis give a mixture of ketones only?
| (i) | CH3 – CH = CH – CH3 |
| (ii) | \[\begin{array}{cc} \ce{CH3 - C - CH = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | 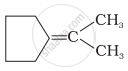 |
| (iv) | \[\begin{array}{cc} \phantom{...................}\ce{CH3}\\ \phantom{..............}/\\ \ce{(CH3)2 C = C}\\ \phantom{..............}\backslash\\ \phantom{...................}\ce{CH3} \end{array}\] |
Concept: undefined >> undefined
In the presence of peroxide addition of HBr to propene takes place according to anti Markovnikov’s rule but peroxide effect is not seen in the case of HCl and HI. Explain.
Concept: undefined >> undefined
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write the IUPAC name of ‘A’.
Concept: undefined >> undefined
An alkene ‘A’ contains three C-C, eight C-H σ bonds and one C-C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write the IUPAC name of ‘A’.
Concept: undefined >> undefined
What is the basic difference between the terms electron gain enthalpy and electronegativity?
Concept: undefined >> undefined
How would you react to the statement that the electronegativity of N on Pauling scale is 3.0 in all the nitrogen compounds?
Concept: undefined >> undefined
