- A homologous series is a group of compounds with the same functional group and similar chemical properties.
- Successive members differ by one CH₂ (methylene) unit, increasing the molecular mass by 14 u.
- All members of a homologous series follow the same general molecular formula (e.g., alkanes CnH2n+2).
- Physical properties such as boiling and melting points show a gradual change with increasing molecular mass.
- Knowing the properties of a few members helps predict the properties of other members in the series.
Definitions [10]
Definition: Organic Chemistry
The branch of chemistry that deals with compounds originally derived from living organisms, such as sugar, starch, proteins, and acetic acid.
or
Organic compounds are the hydrides of carbon (hydrocarbons) and their derivatives. The branch of chemistry dealing with these compounds is called organic chemistry.
Definition: Inorganic Chemistry
The branch of chemistry that deals with compounds obtained from non-living sources or minerals, such as common salt, blue vitriol, and nitrates.
Definition: Homologous Series
The same functional group substitutes for hydrogen in a carbon chain is called a homologous series.
OR
A homologous series is a group of organic compounds having similar structure and similar chemical properties in which the successive compounds differ by a CH2 group.
OR
The series of compounds formed by joining the same functional group in the place of a particular hydrogen atom on the chains having sequentially increasing length is called homologous series.
Define Homologous series.
A series of compounds with similar chemical properties, in which members differ from one another by an additional CH2 group, is called a homologous series.
Definition: Isomers
Compounds having the same molecular formula but different structural formula are known as Isomers and the phenomenon is known as Isomerism. (iso = same, meros = parts).
Definition: Hyperconjugation
The stabilising interaction that involves delocalisation of σ-electrons of C–H bond of an alkyl group linked directly to an atom of unsaturated system or to an atom having unshared p-orbital, is called hyperconjugation.
e.g.
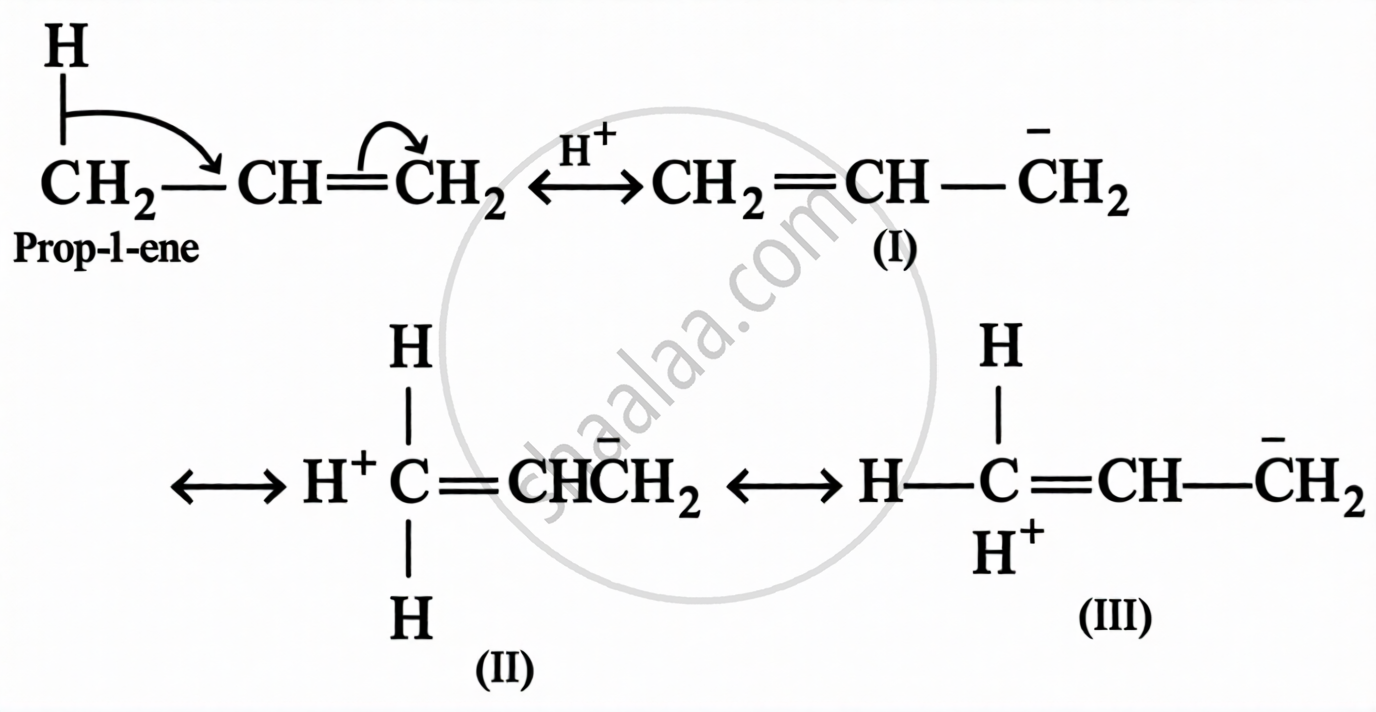
Definition: Sublimation
The change of a solid substance directly into a gas or vapour without first changing into a liquid is called sublimation.
OR
The change from solid state to vapour state without passing through the liquid state is called sublimation, and the substance is said to sublime.
Define sublimation.
The change of state from solid to gas directly is called sublimation.
Define the Stationary phase.
Stationary phase is a solid or a liquid supported on a solid which remains fixed in a place and on which different solutes are adsorbed to a different extent.
Definition: Bond Fission
Bond fission is the process of breaking a covalent bond of an organic compound, also known as bond cleavage.
Key Points
Key Points: Tetravalence of Carbon & Shapes of Organic Compounds
Carbon has 4 electrons in its outermost shell → forms 4 covalent bonds → tetravalent.
Determination of Hybridisation (First Method):
- Count the number of pi (π) bonds on that carbon atom.
- Single-bonded C → sp³ hybridisation
- C with one π bond → sp²
- C with two π bonds → sp
| Bonds on C | Hybridisation | Geometry | Example |
|---|---|---|---|
| 4 sigma, 0 pi | sp³ | Tetrahedral | Methane (CH₄) |
| 3 sigma, 1 pi | sp² | Trigonal planar | Ethene (C₂H₄) |
| 2 sigma, 2 pi | sp | Linear | Ethyne (C₂H₂) |
Shapes of molecules:
- Methane (CH₄) → sp³ hybrid orbitals
- Ethene (C₂H₄) → sp² hybrid orbitals
- Ethyne (C₂H₂) → sp hybrid orbitals
Key Points: Some Characteristic Features of π Bonds
- A π bond is formed by the lateral (side-to-side) overlap of two p orbitals.
- π bonds are weaker than σ bonds (less effective overlap).
- π bonds restrict free rotation around the bond axis — this is why geometrical isomerism exists in alkenes.
- π electrons are more loosely held and easily available for chemical reactions → making alkenes and alkynes more reactive than alkanes.
- Greater the s character of the hybrid orbital, greater the electronegativity.
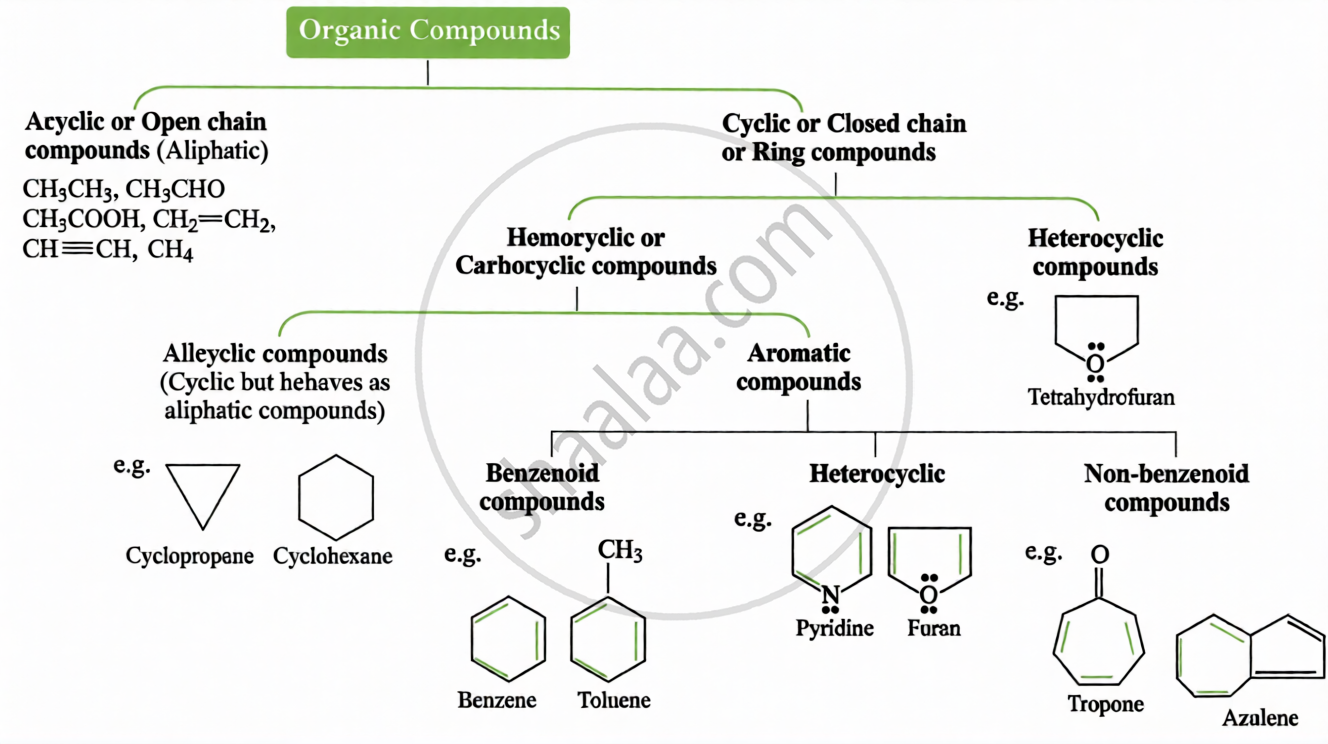
Key Points: Classification of Organic Compounds
(i) Based on Functional Group:
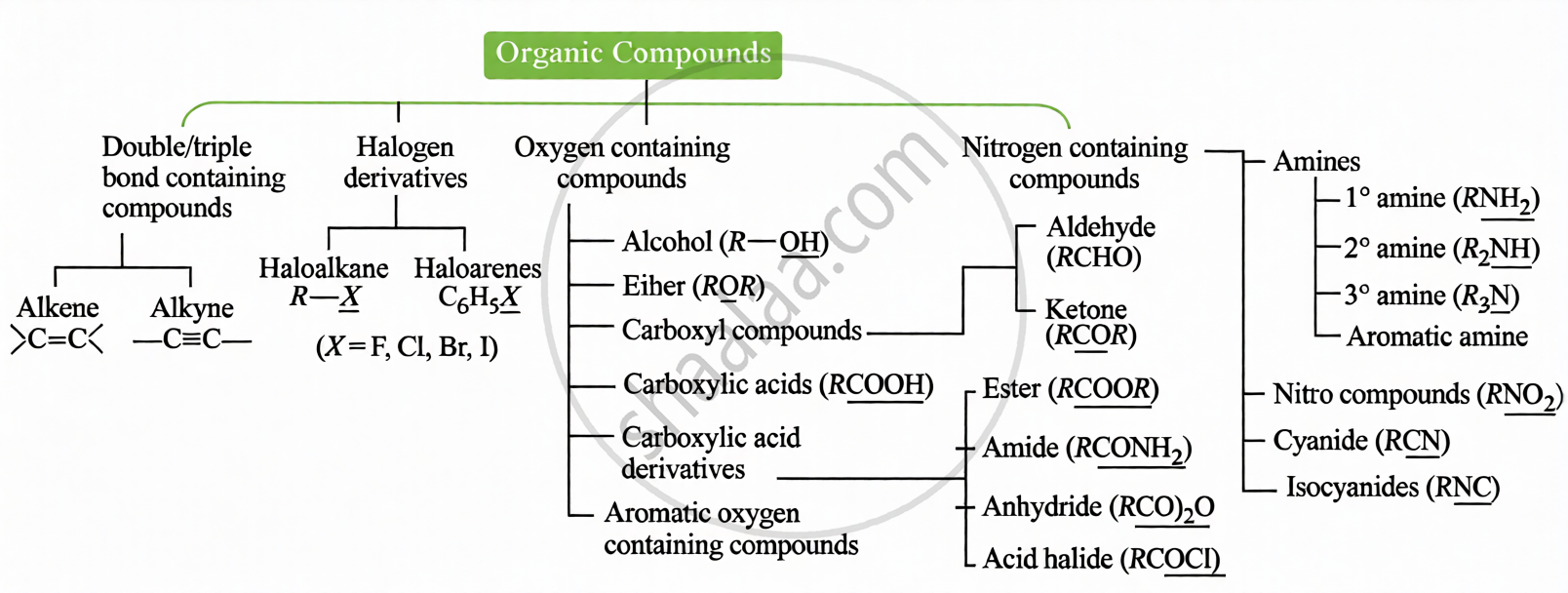
(ii) Based on Carbon Skeleton:
Key Points: Nomenclature of Organic Compounds
IUPAC naming provides a systematic way to name organic compounds based on their structure.
General format:
IUPAC Name = Prefix + Word Root (Stem) + Suffix
- Numbers: Indicate position of substituents
- Prefix: Substituents (alkyl/functional groups)
- Word Root: Number of carbon atoms in longest chain
- Suffix: Principal functional group
IUPAC Rules:
| Rule No. | Rule | Explanation |
|---|---|---|
| Rule 1 | Longest Chain | Select the longest continuous carbon chain as parent chain (not always straight). |
| Rule 2 | Principal Chain Selection | If chains are equal, choose the one with the maximum substituents (PCC). |
| Rule 3 | Numbering | Number chain to give the lowest locants to substituents/functional group. |
| Rule 4 | Functional Group Priority | Principal functional group gets lowest number, then multiple bonds, then substituents. |
| Rule 5 | Multiple Substituents | Use di-, tri-, tetra- for the same groups; these are not considered in alphabetical order. |
| Rule 6 | Alphabetical Order | Different substituents are written in alphabetical order; used to break numbering ties. |
Key Points: Homologous Series
Key Points: Types of Isomerism
1. Structural Isomerism
Same molecular formula but different connectivity (bonding) of atoms.
Types:
- Chain isomerism: Different carbon skeleton (straight/branched)
- Position isomerism: Functional group at different positions
- Functional isomerism: Different functional groups (e.g., alcohol vs ether)
- Metamerism: Different alkyl groups on either side of same functional group
- Tautomerism: Dynamic equilibrium between two forms (keto ↔ enol)
2. Stereoisomerism
Same molecular formula and bonding but different spatial arrangement.
Types:
I. Geometrical isomerism:
- Due to restricted rotation (double bond)
- Forms cis (same side) and trans (opposite side)
II. Optical isomerism:
- Mirror image isomers (enantiomers)
- Show optical activity (rotate plane polarized light)
Key Points: Inductive Effect
- Permanent effect due to shift of σ-electrons
- Electrons move towards more electronegative atom
- Effect decreases with distance (negligible after 3 carbons)
e.g-
![]()
| Type | Meaning | Example |
|---|---|---|
| –I effect | Electron withdrawing group | –NO₂, –Cl |
| +I effect | Electron donating group | –CH₃ |
Key Points: Resonance Effect
Resonance:
- When a single Lewis structure cannot explain all properties of a compound, two or more structures (canonical forms) are written
- The real molecule does not exist as separate forms, it exists as a resonance hybrid
- Resonance hybrid is a weighted average of all canonical forms
- Due to resonance, electrons are delocalised over the molecule
- Resonance hybrid is more stable (lower energy) than any single structure
Resonance Effect (Mesomeric Effect):
| Type | Explanation | Effect on Electron Density | Examples |
|---|---|---|---|
| +R effect | Group donates electrons to conjugated system | Increases electron density at certain positions | –OH, –OR, –NH₂, –NHR |
| –R effect | Group withdraws electrons from conjugated system | Decreases electron density at o- and p-positions | –NO₂, –CN, –CHO, –COOH |
Key Points: Electromeric Effect
A temporary effect involving the complete transfer of shared π-electrons to one of the atoms in a multiple bond, in the presence of an attacking reagent.
| Type | Direction of π-electron transfer | Example |
|---|---|---|
| +E Effect | π electrons transferred to the atom to which the reagent gets attached | |
| –E Effect | π electrons transferred to the atom to which the reagent does not attach | 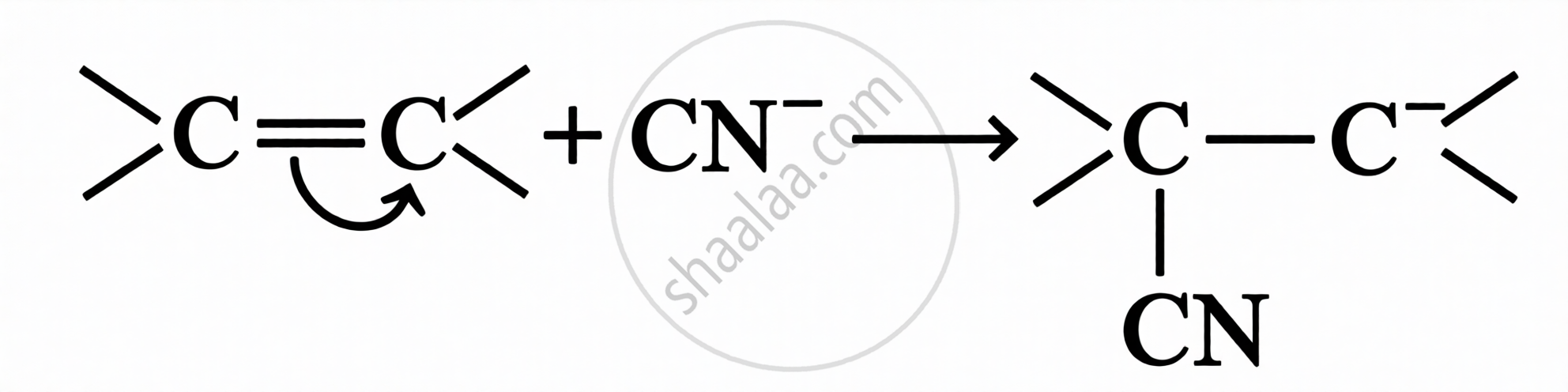 |
Key Points: Hyperconjugation
Also called the Baker-Nathan effect or no-bond resonance.
Conditions for hyperconjugation:
- The compound must have at least one sp²-hybridised carbon (alkene, alkyl carbocation, or alkyl free radical)
- The α-carbon must have at least one H–C bond attached to the unsaturated system
- More the number of H–C bonds at α-carbon → greater the stabilisation → more stable the alkene/carbocation
Resonance vs. Hyperconjugation:
| Feature | Resonance | Hyperconjugation |
|---|---|---|
| Electrons involved | π electrons delocalised | σ electrons of C–H bond delocalised |
| Stability provided | Greater | Lesser |
| Bond type | π bond | σ bond |
Key Points: Crystallisation Method
- Based on difference in solubility of the compound and impurities at different temperatures.
- Compound more soluble in hot solvent → dissolved at high temperature → cooled → pure crystals form → impurities remain in solution (mother liquor).
- Common solvents: water, alcohol, ether, chloroform, acetone, benzene, petroleum ether.
- Example: Benzoic acid (slightly soluble in cold water, completely soluble in hot water) → separated from naphthalene.
- Fractional crystallisation: Used when two compounds have different solubilities in the same solvent → less soluble crystallises first on cooling (e.g., K₂CO₃ and KCl).
Key Points: Fractional Distillation Method
- Used for separation of a mixture of two or more miscible liquids with close boiling points (difference < 25°C).
- Uses a fractionating column for repeated vaporisation and condensation.
- Distillate collected in fractions at different temperatures.
- Example: Acetone (b.p. 330 K) and methyl alcohol (b.p. 338 K); benzene and toluene.
- Cannot be used to separate azeotropic mixtures (constant boiling point mixtures).
Key Points: Steam Distillation
Applicable for separation/purification of organic compounds (solids or liquids) that are:
- Insoluble in water
- Volatile in steam
- Have vapour pressure 10–15 mm Hg at 373 K
- Contain non-volatile impurities
Organic compound is passed with steam → distils at a temperature below its normal boiling point.
Example: Aniline (b.p. 457 K) purified by steam distillation at 371.5 K.
Other examples: nitrobenzene, bromobenzene, o-nitrophenol, salicylaldehyde, o-hydroxyacetophenone, essential oils, turpentine oil.
Key Points: Chromatography Method
- Introduced by Tswett in 1906.
- Based on the difference in rates at which different components of a mixture move through a porous medium (stationary phase) under the influence of a solvent or gas (mobile or moving phase).
- The technique separates a mixture by distributing its components between one stationary phase and one mobile phase.
- Mobile phase determines the Rᶠ or R factor — dependent on the solubility of components in the given eluent.
Key Points: Qualitative Analysis of Organic Compounds
| Element | Method / Test | Principle | Observation | Inference |
|---|---|---|---|---|
| Carbon (C) | Copper oxide test | C oxidised to CO₂ by CuO | CO₂ turns lime water milky | Carbon present |
| Hydrogen (H) | Copper oxide test | H oxidised to H₂O by CuO | Anhydrous CuSO₄ turns blue | Hydrogen present |
| Nitrogen (N) | Lassaigne's test | Na + C + N → NaCN → Prussian blue with FeSO₄ + FeCl₃ | Prussian blue colour | Nitrogen present |
| Nitrogen + Sulphur (N+S) | Lassaigne's test | SCN⁻ formed instead of CN⁻ and S²⁻ separately | Blood red colour with FeCl₃ (Fe(SCN)²⁺) | Both N and S present |
| Sulphur (S) only | Sodium nitroprusside test | Na₂S + [Fe(CN)₅NO]²⁻ → coloured complex | Violet colour | Sulphur present |
| Chlorine (Cl) | Lassaigne's test + AgNO₃ | NaCl + AgNO₃ → AgCl↓ | White ppt, soluble in NH₄OH | Chlorine present |
| Bromine (Br) | Lassaigne's test + AgNO₃ | NaBr + AgNO₃ → AgBr↓ | Pale yellow ppt, partially soluble in NH₄OH | Bromine present |
| Iodine (I) | Lassaigne's test + AgNO₃ | NaI + AgNO₃ → AgI↓ | Yellow ppt, insoluble in NH₄OH | Iodine present |
| Halogen (Cl/Br) | Beilstein test | Organic compound on Cu wire in Bunsen flame | Green/blue-green flame | Halogen (Cl or Br) present |
| Phosphorus (P) | Ammonium molybdate test | P oxidised to phosphate → ammonium phosphomolybdate | Canary yellow ppt | Phosphorus present |
Key Points: Types of Bond Fission
| Feature | Homolytic Fission (Homolysis) | Heterolytic Fission (Heterolysis) |
|---|---|---|
| Meaning | Bond breaks equally | Bond breaks unequally |
| Electron Distribution | Each atom gets one electron | One atom gets both electrons |
| Species Formed | Free radicals (A•, B•) | Ions (A⁺, B⁻) |
| Conditions | UV light, heat, peroxides | Polar medium, electronegativity difference |
| Example | Cl₂ → Cl• + Cl• | HCl → H⁺ + Cl⁻ |
Concepts [40]
- Organic Chemistry
- Tetravalence of Carbon - Shapes of Organic Compounds
- Some Characteristic Features of π Bonds
- Structural Representation of Organic Compounds
- Complete, Condensed and Bond-line Structural Formulas
- Three-dimensional Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature of Organic Compounds
- Homologous Series
- The IUPAC System of Nomenclature
- IUPAC Nomenclature of Alkanes
- Nomenclature of Organic Compounds having Functional Group(s)
- Nomenclature of Substituted Benzene Compounds
- Isomerism
- Fission of a Covalent Bond
- Substrate and Reagent
- Electron Movement in Organic Reactions
- Electron Displacement Effects in Covalent Bonds
- Inductive Effect
- Resonance Structure
- Resonance Effect
- Electromeric Effect (E Effect)
- Hyperconjugation
- Types of Organic Reactions and Mechanisms
- Methods of Purification of Organic Compounds
- Sublimation
- Crystallisation Method
- Fractional Distillation Method
- Steam Distillation
- Differential Extraction
- Chromatography Method
- Qualitative Analysis of Organic Compounds
- Qualitative Analysis of Organic Compounds - Detection of Other Elements
- Quantitative Analysis of Carbon and Hydrogen
- Quantitative Analysis of Nitrogen
- Quantitative Analysis of Halogens
- Quantitative Analysis of Sulphur
- Quantitative Analysis of Phosphorus
- Quantitative Analysis of Oxygen
- Bond Fission
