(English Medium)
Academic Year: 2025-2026
Date & Time: 11th March 2026, 11:00 am
Duration: 2h
Advertisements
Time allowed: Three hours
- Answers to this Paper must be written on the paper provided separately.
- You will not be allowed to write during first 15 minutes.
- This time is to be spent in reading the question paper.
- The time given at the head of this Paper is the time allowed for writing the answers.
- Section A is compulsory. Attempt any four questions from Section B.
- The intended marks for questions or parts of questions are given in brackets [].
Instruction for the Supervising Examiner
Kindly read aloud the Instructions given above to all the candidates present in the Examination Hall.
A non-metal which reacts with concentrated sulphuric acid to form two gases which turn lime water milky is ______.
Sulphur
Carbon
Oxygen
Nitrogen
Chapter:
Which of the following clement pairs will form an ionic bond?
| Pair | |
| P | Elements of Group 1 & Group 2 |
| Q | Elements of Group 14 & Group 16 |
| R | Elements of Group 2 & Group 17 |
| S | Elements of Group 15 & Group 18 |
P
Q
R
S
Chapter:
The electronic configuration of an element is 2, 8, 2. The hydroxide of this element can produce ______ hydroxyl ions per molecule.
3
2
1
0
Chapter:
With respect to the electrolysis of copper (II) sulphate solution using copper electrodes, which statement is correct?
Copper metal is deposited at the negative electrode.
Oxygen gas is produced at the positive electrode.
The positive electrode increases in mass.
The negative electrode decreases in mass.
Chapter:
Identify the equation that shows the reaction of ethane with chlorine in the presence of ultraviolet light.
\[\ce{C2H6 + Cl2 -> C2H6Cl2}\]
\[\ce{C2H6 + Cl2 -> C2H4Cl2 + H2}\]
\[\ce{C2H6 + Cl2 -> C2H5Cl +HCl}\]
\[\ce{C2H6 + Cl2 -> 2CH3Cl}\]
Chapter:
A hydrocarbon X undergoes the reaction under suitable conditions as shown below:
\[\begin{array}{cc}
\ce{\phantom{.....}H\phantom{....}H\phantom{....}H\phantom{....}H\phantom{.............}H\phantom{.......}H\phantom{....}H\phantom{...................}H\phantom{.....}}\\
\phantom{.....}|\phantom{......}|\phantom{......}|\phantom{......}|\phantom{...............}\backslash\phantom{.......}|\phantom{.....}|\phantom{.....................}|\phantom{.....}\\
\ce{H - C - C - C - C - H -> C = C - C - H + H - C - H}\\
\phantom{......}|\phantom{......}|\phantom{......}|\phantom{......}|\phantom{...............}/\phantom{.............}|\phantom{.....................}|\phantom{......}\\
\ce{\underset{\phantom{........}(X)}{\phantom{......}H\phantom{....}H\phantom{....}H\phantom{....}H}\underset{\phantom{.................}(Y)}{\phantom{............}H\phantom{..............}H}\phantom{...................}\underset{(Z)}{H}\phantom{......}}
\end{array}\]
Which compound(s) is/are unsaturated?
X only
Y only
X and Z
Y and Z
Chapter:
Rita added dilute hydrochloric acid to four metals. She recorded her observations in the table given below. While noting her observations she made some errors.
| Metals | Observations | |
| 1 | copper | a gas was given off |
| 2 | iron | a gas was given off |
| 3 | magnesium | no a gas was given off |
| 4 | zinc | a gas was given off |
Which observation(s) made by Rita is/are correct?
1 only
3 only
Both 1 and 4
Both 2 and 4
Chapter:
An atom of X forms an ion according to the equation X → X+2.
The atomic number of the atom X is:
16
10
12
14
Chapter:
Salts can be prepared by the action of dilute acid on:
- bases
- carbonates
- metals
- sulphites
The method which cannot be used for the preparation of copper salts is:
1
2
3
4
Chapter:
The compound that has the highest melting point amongst the following is ______.
Methane
Sodium chloride
Ammonia
Ethanol
Chapter:
Assertion (A): Dilute Sulphuric acid is a stronger electrolyte than concentrated Sulphuric acid.
Reason (R): Dilute Sulphuric acid has a higher concentration of mobile ions.
(A) is true but (R) is false.
(A) is false but (R) is true.
Both (A) and (R) are true and (R) is the correct explanation of (A).
Both (A) and (R) are true, but (R) is not the correct explanation of (A).
Chapter:
What volume of carbon dioxide is produced at STP when 5 litres of propane is burnt completely according to the equation given below?
\[\ce{C3H8 + 5O2 -> 3CO2 + 4H2O}\]
10 litres
15 litres
20 litres
25 litres
Chapter:
An unsaturated hydrocarbon with three atoms of carbon and six atoms of hydrogen is ______.
propyne
propane
propene
propanol
Chapter:
Assertion (A): In the electrolysis of acidified water, the volume of hydrogen liberated is twice the volume of oxygen formed.
Reason (R): Water has hydrogen and oxygen in the ratio of 1 : 2 by volume.
(A) is true but (R) is false.
(A) is false but (R) is true.
Both (A) and (R) are true, and (R) is the correct explanation of (A).
Both (A) and (R) are true, but (R) is not the correct explanation of (A).
Chapter:
In the electrolysis of molten aluminium oxide for extraction of aluminium, the following reactions take place:
- \[\ce{Al^3+ + 3e^- -> Al}\]
- \[\ce{2O^2- -> O2 + 4e^-}\]
- \[\ce{C + O2 -> CO2}\]
- \[\ce{Al2O3 ⇌ 2Al^3+ + 3O^2-}\]
The reaction that takes place at the negative electrode is:
1
2
3
4
Chapter:
Given below is the industrial process for the manufacture of ammonia gas.
Study the schematic diagram to answer the following questions.
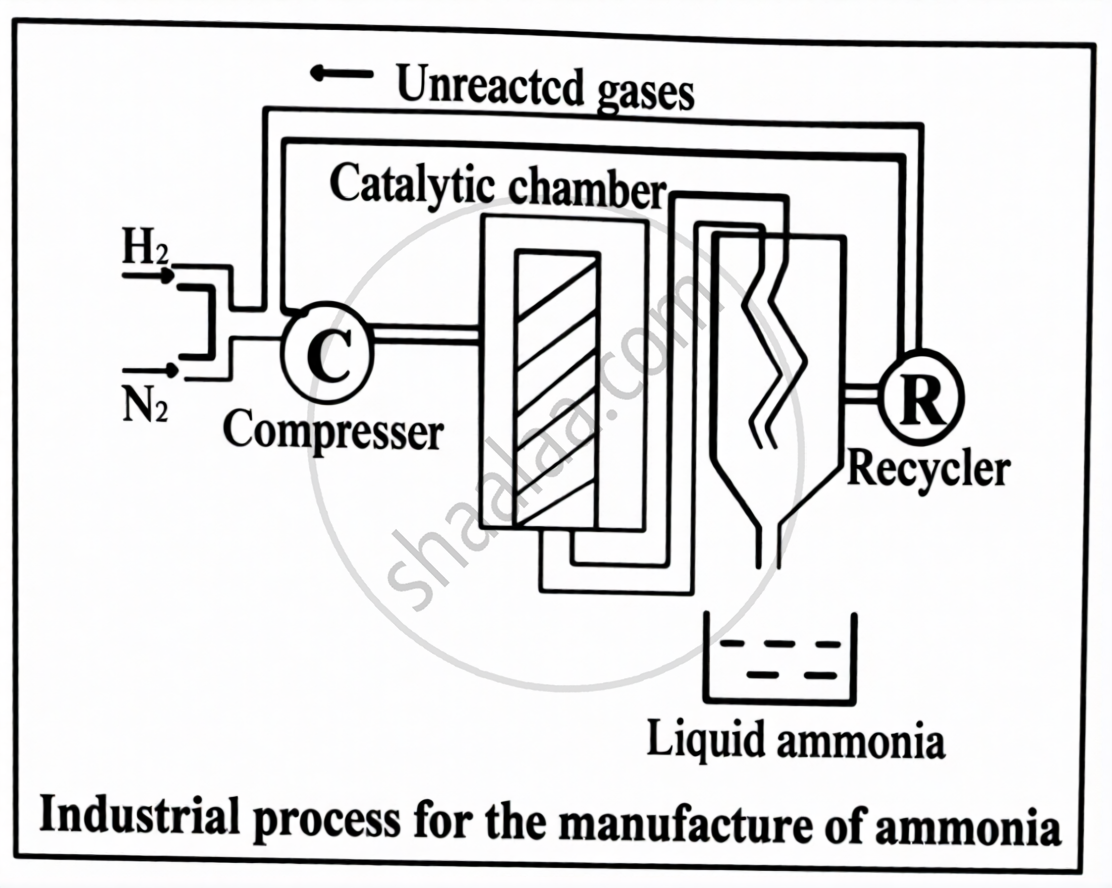
- Name the process.
- Which catalyst is used in the above process?
- In the above diagrammatic setup, how is ammonia gas separated from the unreacted gases to obtain liquid ammonia?
- Which two properties of ammonia gas can be demonstrated by the Fountain Experiment?
Chapter:
Identify the following term/compound:
A compound which reacts with water to give acetylene gas.
Chapter:
Identify the gas evolved when sodium propionate is heated with sodalime
Chapter: [12.1] Organic Compounds
Identify the following term/compound:
A reddish-brown precipitate formed when ferric chloride solution reacts with an alkali.
Chapter:
Identify the following term/compound:
A pair of electrons present in an atom which is not shared with any other atom during bond formation.
Chapter:
Identify the following term/compound:
The relative molecular mass of a substance expressed in grams.
Chapter:
Advertisements
Match Column A with Column B:
| Column A | Column B | ||
| (a) | \[\ce{CuO + C -> Cu + CO}\] | 1. | Oxidation |
| (b) | \[\ce{Al^3+ + 3e^- -> Al}\] | 2. | lonizaon |
| (c) | \[\ce{PbBr2 -> Pb^2+ + 2Br^1-}\] | 3. | Reduction |
| (d) | \[\ce{2O^2- - 2e^- -> O2}\] | 4. | Redox |
| (e) | \[\ce{HCl -> H^+ + Cl^-}\] | 5. | Electrolytic dissociation |
Chapter:
The oxide that dissolves in caustic potash (KOH) is ______.
ZnO
CuO
Chapter:
The reaction that takes place at the anode during the electrolysis of molten lead bromide is ______.
\[\ce{2Br - 2e^- -> 2Br^-}\]
\[\ce{2Br^- - 2e^- -> Br2}\]
Chapter:
The volume occupied by 8 grams of oxygen gas at STP is ______ litres. [Atomic weight of O = 16]
5.6
8.96
Chapter:
______ does not give hydrogen gas when it reacts with cold and very dilute nitric acid.
Cu
Mn
Chapter:
Give the IUPAC name for the following organic compound:
\[\begin{array}{cc}
\ce{HO - CH - CH2 - CH3}\\
|\phantom{...............}\\
\ce{CH3\phantom{...........}}
\end{array}\]
Chapter:
Give the IUPAC name for the following organic compound:
\[\begin{array}{cc}
\ce{H3C - CH2 - CH - CH2 - CH3}\\
|\phantom{..}\\
\ce{\phantom{..}CH3}
\end{array}\]
Chapter:
Draw the structure for the following compound:
Butanal
Chapter:
Draw the structural diagram for the following organic compound:
pent-2-yne
Chapter:
Draw the structural formula for the following compound:
Isomer of n-butane
Chapter:
|
Alex was given a solution of an unknown salt Y for analysis. He performed the following tests and recorded his observations:
|
Based on the observations made by Alex, identify:
- the anion and
- the cation present in salt Y.
Chapter:
Study the reaction given below and answer the question that follows:
\[\ce{NaNO3 + \underset{Conc.}{H2SO4} ->[< 200° C][]NaHSO4 + HNO3}\]
Give one reason for maintaining the temperature below 200°C.
Chapter:
Study the reaction given below and answer the question that follows:
\[\ce{NaNO3 + \underset{Conc.}{H2SO4} ->[< 200° C][]NaHSO4 + HNO3}\]
Why is concentrated Sulphuric acid used in the above reaction?
Chapter:
Element A belongs to group 15 and period 2 of the Periodic Table.
- Identify the element A.
- Write the formula of the compound formed when element A combines with hydrogen.
- Draw the dot (•) and cross (x) structure of the compound formed in (b) above.
Chapter:
Complete and balance the following equation:
\[\ce{C2H4Br2 + alcoholic KOH ->[\Delta]}\]
Chapter:
Complete and balance the following equation:
\[\ce{NH3 (excess) + Cl2 ->}\]
Chapter:
Complete and balance the following equation:
\[\ce{C12H22O11 ->[(conc{.}) H2SO4][]}\]
Chapter:
Differentiate between the following pairs based on the criteria given:
Acetic acid and Sulphuric acid (number of replaceable hydrogen ion per molecule)
Chapter:
Differentiate between the following pairs based on the criteria given:
Electrolyte and Metallic conductor (the particles conducting electricity)
Chapter:
Equal volumes of ammonia gas and chlorine gas are kept in two different containers under the same conditions of temperature and pressure. Find the number of molecules contained in chlorine gas when the mass of ammonia is 34 g. [Atomic weight: C1 = 35.5, H = 1, N = 14]
Chapter:
Advertisements
|
Elements P, Q, and R are in the same period of the modern periodic table.
|
Answer the following questions based on the above information:
- Which clement would be most difficult to reduce among P, Q and R?
- Which element is expected to have the smallest atomic radius among P, Q and R?
- Arrange P, Q and R in order of decreasing ionization potential.
Chapter:
Given below are some chemicals.
| Lead nitrate, Copper, Hydrochloric acid, Lead oxide, Iron, Sodium carbonate, Copper carbonate, Chlorine |
Using suitable chemicals from the box given above, write balanced chemical equations for the preparation of the following salts.
(Note: The chemicals may be used more than once, if needed.)
- Lead (II) carbonate
- Copper (II) chloride
- Iron (II) chloride
Chapter:
| An unsaturated hydrocarbon having two carbon atoms was made to react with two moles of hydrogen gas in the presence of a catalyst to give a saturated hydrocarbon. |
Using the above information, answer the following questions:
- The type of reaction that occurred is ______. (addition/substitution)
- Name a suitable catalyst which is used in the reaction.
- Write a balanced chemical equation for the above reaction.
Chapter:
During the manufacture of sulphuric acid, a chemist collected the gas formed after catalytic oxidation of sulphur dioxide.
- Name the gas collected.
- Which acid is formed when the gas collected by the chemist is dissolved directly in water?
- Why should we not dissolve this gas directly in water?
Chapter:
The diagram given below shows the electroplating of a spoon with silver. With reference to the diagram, answer the following questions:
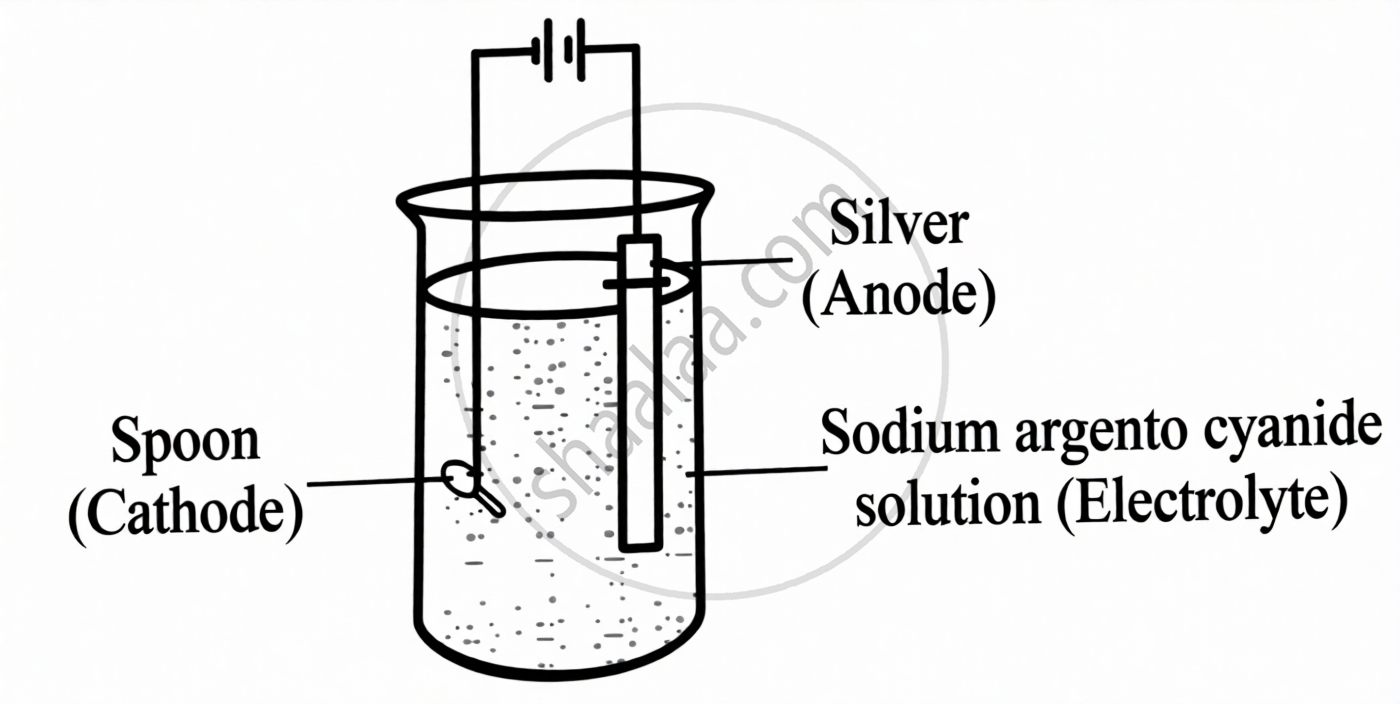
- Write an equation for the reaction that occurs at the cathode.
- Silver nitrate solution is not preferred as an electrolyte in the above process. Give a reason.
- Why is alternating current not used in the process?
- What is the observation at the anode?
Chapter:
Give reasons why hydrochloric acid cannot form an acid salt.
Chapter:
Give reasons why electronegativity increases across a period.
Chapter:
State whether the following statement is True or False:
Organic compounds having different molecular formula, but same structural formula are called isomers.
Chapter:
State whether the following statement is True or False:
A salt is a compound formed by partial or complete replacement of the hydrogen ion of an acid by a metal or electro positive ion.
Chapter:
Anil is a laboratory assistant in a college. He prepared nitric acid but stored it in a transparent container exposed to sunlight. After a few days, brown fumes were observed in the container and the acid turned slightly yellowish in colour.
- What was the error made by Anil while storing the acid?
- Why were brown fumes observed in the container?
- How can the yellowish tinge in the acid be removed?
Chapter:
\[\ce{Zn + 4HNO3 -> Zn(NO3)2 + 2H2O + 2NO2}\]
32.5 g of zinc reacts with concentrated nitric acid as given in the above equation.
- How many moles of zinc was required in the reaction?
- Find the mass of nitric acid needed to react with 32.5 g of zinc.
- Find the volume of nitrogen dioxide liberated in (b).
[Atomic weight: H = 1, N = 14, O = 16, Zn = 65]
Chapter:
Carbon reacts with an acid to form carbon dioxide, water and nitrogen dioxide.
- Name the acid used in the reaction.
- Write a balanced chemical equation for the reaction that occurs.
Chapter:
|
Bauxite is the principal ore used in the commercial extraction of aluminium. The Bayer’s process is used to refine bauxite into pure alumina, with caustic soda playing a crucial role in the initial stage of the process. |
Based on this, answer the following questions:
- Explain the reason behind the addition of caustic soda during the Bayer’s process.
- Write a balanced chemical equation representing the reaction between bauxite and caustic soda during the Bayer’s process.
Chapter:
Give one relevant observation for the following:
Sodium hydroxide is added dropwise to Calcium nitrate solution.
Chapter:
Give one relevant observation for the following:
Dilute Hydrochloric acid is added to Iron (II) sulphide.
Chapter:
Give one relevant observation for the following:
An amphoteric metal is added to hot concentrated alkali.
Chapter:
Study the reaction scheme shown below and identify the reactants A, B and C.
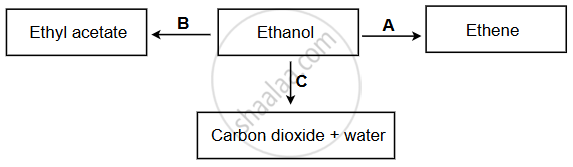
Chapter:
5.6 litres of gas Z weighs 32 g at STP. What is the molecular weight and vapour density of Z?
Chapter:
Name the following:
The most electronegative element of Period 2.
Chapter:
Name the following:
The largest atom of Period 3.
Chapter:
Given below are a few elements.
| Li, K, Ca, F |
Identify the element which:
- has the least atomic radius.
- has two valence electrons.
- is the most electropositive.
Chapter:
K, L and M are beakers containing three different solutions as shown below.

Name the beaker which:
- contains only ions.
- contains only molecules.
- has pH less than 7.
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ICSE Class 10 Chemistry with solutions 2025 - 2026
Previous year Question paper for CISCE ICSE Class 10 -2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ICSE Class 10 .
How CISCE ICSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
