Science (English Medium)
Academic Year: 2025-2026
Date: मार्च 2026
Advertisements
GENERAL INSTRUCTIONS:
Read the following instructions carefully.
- There are 33 questions in this question paper with internal choice.
- SECTION A consists of 16 multiple-choice questions carrying 1 mark each.
- SECTION B consists of 5 short answer questions carrying 2 marks each.
- SECTION C consists of 7 short answer questions carrying 3 marks each.
- SECTION D consists of 2 case-based questions carrying 4 marks each.
- SECTION E consists of 3 long answer questions carrying 5 marks each.
- All questions are compulsory.
- Use of log tables and calculators is not allowed.
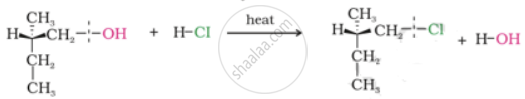
Which of the following reactions will lead to the formation of ethyl methyl ketone?
Heating CH3CH(CH3)CH2OH with acidified Na2Cr2O7.
Passing CH3C(OH)CH3 over heated copper.
Ozonolysis of CH3CH2C(CH3) = CHCH3
Acetylene on reaction with HgSO4/H2SO4.
Chapter:
Consider the reaction and identify B and C:
\[\ce{CH3CH2CH2CH2Cl ->[NaOH + Ethanol] A}\]
\[\ce{A ->[H2O, H^+] B}\] and \[\ce{A ->[(i) B2H6 (ii) H2O2, OH^-]C}\]
B = C = Butanol
B = Butanol, C = Butene
B = Butan-2-ol, C = Butanol
B = Butene, C = Butan-2-ol
Chapter:
The counter ion in the coordination compound [Co(NH3)5(NO2)]Cl2 is ______.
Ammine
Cobalt
Chloride
Nitro
Chapter:
The organic compounds A, B and C are amines having equivalent molecular weight. A and B on reaction with benzene sulphonyl chloride give a white precipitate; however, the white precipitate obtained from compound B remains insoluble in NaOH.
The variation in the boiling point of A, B and C can be seen as:
A > B > C
B > A > C
A = B > C
C > B > A
Chapter:
70 gm solute is dissolved in 700 gm solvent to prepare a solution having density 1.5 g/ml. The ratio of its molality and molarity will be ______.
0.77
1.4
0.73
1.3
Chapter:
Match the column I and column II:
| Column I | Column II | ||
| A. |
|
(i) | Additional reaction |
| B. | 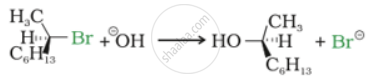 |
(ii) | Elimination reaction |
| C. |
\[\begin{array}{cc} |
(iii) | SN2 reaction |
| D. | \[\ce{CH3CH = CH2 + H-I ->H3CH2CH2I + CH3CHICH3}\] | (iv) | SN1 reaction |
A - (i), B - (ii), C - (iii), D - (iv)
A - (iv), B - (ii), C - (iii), D - (i)
A - (i), B - (iii), C - (ii), D - (iv)
A - (iv), B - (iii), C - (ii), D - (i)
Chapter:
In o-cresol, the –OH group is attached to the carbon that is ______.
sp3 hybrid
sp2 hybrid
sp hybrid
dsp2 hybrid
Chapter:
Which of the following is laevorotatory in nature?
alpha-D-glucose
beta-D-glucose
beta-D-fructose
sucrose
Chapter:
The name inner transition metals is often used to refer to ______.
lanthanoids
actinoids
both lanthanoids and actinoids
d-block elements
Chapter:
\[\ce{\Lambda^{\circ}_m CH3COOH}\] can be calculated if the values of the following are given:
- \[\ce{\Lambda^{\circ}_m HCl, \Lambda^{\circ}_m KCl and \Lambda^{\circ}_m CH3COOK}\]
- \[\ce{\Lambda^{\circ}_m NaCl, \Lambda^{\circ}_m KCl and \Lambda^{\circ}_m CH3COONa}\]
- \[\ce{\Lambda^{\circ}_m H2SO4, \Lambda^{\circ}_m Na2SO4 and \Lambda^{\circ}_m CH3COONa}\]
Only 1
Either 1 or 2
Either 1 or 3
Either 2 or 3
Chapter:
Which of the following will give a yellow or orange ppt. with 2,4-DNP?
- Propanal
- Propanone
- Propanoic acid
(i) and (ii)
(ii) and (iii)
(i) and (iii)
(i), (ii) and (iii)
Chapter:
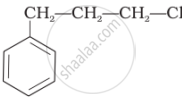
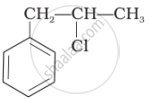
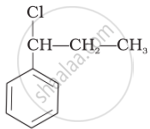
Identify the secondary benzylic halide and primary alkyl halide from the following:
(i) and (iii)
(iv) and (ii)
(iii) and (iv)
(i) and (ii)
Chapter:
Assertion (A): Secondary aliphatic amines react with nitrous acid to form aliphatic diazonium salts which being unstable, liberate nitrogen gas quantitatively.
Reason: Nitrogen gas evolved on reaction with nitrous acid can be used for the estimation of proteins and amino acids.
Select the most appropriate answer from the options given below:
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
Assertion (A): Care should always be taken to ensure that solutions that flow in the bloodstream are of the same osmotic pressure as that of the blood.
Reason (R): Sodium ions and potassium ions are responsible for maintaining proper osmotic pressure balance inside and outside of the cells of an organism.
Select the most appropriate answer from the options given below:
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
Assertion (A): Starch forms a colloidal solution with water.
Reason (R): Starch contains 80-85% of amylopectin, which is insoluble in water.
Select the most appropriate answer from the options given below:
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
Assertion (A): Secondary cells are used in inverters.
Reason (R): A primary cell can be recharged by passing current through it in the opposite direction after it has been used.
Select the most appropriate answer from the options given below:
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
When 50 mL of phenol and 50 mL of aniline are mixed, predict whether the volume of the solution is equal to, greater than or less than 100 mL. Give a reason to support your answer.
Chapter:
Ritesh suggested adding salt to the box containing ice. He said this would keep the cold drink bottles cold for a longer time. How will Ritesh justify his suggestion?
Chapter:
BaCl2 on reaction with Na2SO4 in aqueous solution gives white precipitate. If the two solutions are separated by a semi-permeable membrane, will there be appearance of a white precipitate due to osmosis?
Chapter:
Why does water stop boiling when sugar is added to boiling water?
Chapter:
Consider the graph for the reaction:
\[\ce{H2 + I2 {⇌} 2HI}\]

- Calculate enthalpy of reaction and activation energy for the backward reaction.
- How will the catalyst affect the rate of this reaction? Explain.
Chapter:
Carry out the following conversion:
Nitrobenzene to 4-bromobenzenamine.
Chapter:
Carry out the following conversion:
Chlorophenylmethane to 2-phenyl-ethanamine.
Chapter:
Write the formula of the following coordination complex:
Diaquasilver(I) dichloridoargentate(I)
Chapter:
Advertisements
Write the formula of the following coordination complex:
Dihydroxidobis(triphenylphosphine)nickel(II)
Chapter:
The mechanism of formation of alcohols from alkenes is given below. Rectify the errors in the mechanism and rewrite the corrected steps:
| Step 1 | 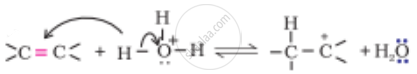 |
| Step 2 | 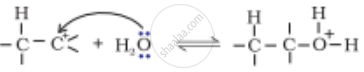 |
| Step 3 | 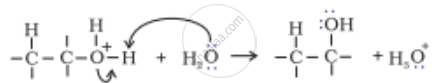 |
Chapter:
What will be the vapour pressure of a 1 molal aqueous solution of MgCl2, assuming dissociation of MgCl2 to be 70 mole percent?
(Vapor pressure of pure water at 25°C is 23.8 mm Hg.)
Chapter:
Write the Nernst equation for the following:
\[\ce{Ni_{(s)} + Cu^2+_{ (aq)} -> Ni^2+_{ (aq)} + Cu_{(s)}}\]
Chapter:
Write the Nernst equation for the following:
\[\ce{Al_{(s)} + FeSO4_{(aq)} -> Al2(SO4)3_{(aq)} + Fe_{(s)}}\]
Chapter:
Write the Nernst equation for the following:
\[\ce{Mg_{(s)} + Mg^2+_{ (aq)}//Ag^+_{ (aq)}/Ag_{(s)}}\]
Chapter:
Explain the following:
Toluene on treatment with Cl2 in sunlight gives benzyl chloride, whereas when treated with Cl2 in dark gives o-chlorobenzene and p-chlorobenzene.
Chapter:
Explain the following:
Finkelstein reaction is carried out in the presence of dry acetone.
Chapter:
Explain the following:
Neopentyl chloride has a lower boiling point than isopentyl chloride.
Chapter:
Which of the following elements will exhibit similar magnetic behaviour and why?
Magnesium (Atomic No. 12), Chromium (Atomic No. 24), Iron (Atomic No. 26) and Molybdenum (Atomic No. 42).
Chapter:
Which of the following elements will form white salts and why?
Zinc (Atomic No. 30), Scandium (Atomic No. 21), Nickel (Atomic No. 28) and Vanadium (Atomic No. 23).
Chapter:
Arrange the products obtained in the following cases in the increasing order of their pKa values:
- Oxidation of ethanol in the presence of acidified potassium dichromate.
- Reaction of propanoic acid with Br2 in the presence of red phosphorus.
- Reaction of isopropyl magnesium bromide with carbon dioxide, followed by hydrolysis.
- Reaction of propanoic acid with Cl2 in the presence of red phosphorus.
Chapter:
Carry out the following conversion.
Butan-2-one to 3-Methylpentan-3-ol.
Chapter:
Carry out the following conversion.
Anisole to 4-Methoxytoluene.
Chapter:
How is phenol converted into the following?
Benzene
Chapter: [7] Alcohols, Phenols and Ethers
Carry out the following conversion:
Chloroethane to Ethoxyethane.
Chapter:
Are the enthalpies of atomisation of Zinc and Copper matched correctly? Justify your answer.
| Element | Enthalpy of atomisation/kJ mol−1 |
| Zinc | 339 |
| Copper | 130 |
Chapter:
Out of sulphuric acid and hydrochloric acid, which acid will you prefer for permanganate titrations and why?
Chapter:
|
Conductivity of Aqueous Solutions:
Electrical conductivity is based on the flow of ions. Slightly ionized substances are weak electrolytes. Weak acids and bases would be categorized as weak electrolytes because they do not completely dissociate in solution. Highly ionized substances are strong electrolytes. Strong acids and salts are strong electrolytes because they completely ionize in solution. The ions carry the electric charge through the solution, thus creating an electric current. The current, if sufficient enough, will light one or both LEDs on a conductivity meter, shown at right. The meter has a 9V battery, two parallel copper electrodes and two LEDs, one green and one red. The conductivity of a solution can be tested by switching the meter on and dipping the copper electrodes in the solution to be tested. Substances that do not conduct an electric current are called non-electrolytes. Non-electrolytes do not ionize; they do not contain moveable ions. The LEDs of a conductivity meter will not light because there are no ions to carry the electric current. The table given below is a guide to the possible conductivity measurements.
|
Based on the information provided above, answer the following questions:
I. Is it possible to identify whether the given solution is 1 M NaOH or 1 M HCl using the conductivity meter? Justify your answer. (1)
II. What is the possible pH value of the solution if the glow of the green LED is medium and the red LED glows very brightly? (1)
(i) 1 (ii) 13 (iii) 5 (iv) 8
- (i) and (ii)
- (i) and (iii)
- (ii) and (iv)
- (iii) and (iv)
OR
Write down the observations if the conductivity meter is dipped in distilled water.
III. Refer to the table given below and draw the molar conductivity vs. concentration curve for solutions A and B. (2)
| Solution | Red LED | Green LED |
| A | bright | dim |
| B | dim | off |
Chapter:
|
Iron-EDTA complex in food fortification:
Food fortification is defined as the practice of adding vitamins and minerals to commonly consumed foods during processing to increase their nutritional value. It is a proven, safe and cost-effective strategy for improving diets and the prevention and control of micronutrient deficiencies. A food product (such as rice, wheat flour, edible oil) that is fortified through the addition of fortificants is called a “vehicle”. In African and south Asian countries, 40% of the population suffers from anaemia. Average human needs nearly 10mg of iron daily. Iron fortification may be useful in fighting iron deficiencies in humans. Reduced iron and several iron salts have been used in the past as iron fortification; however, not all are suitable for this purpose in terms of iron absorption. Recent studies have shown that beverages containing sugar fortified with either Ferrous sulphate or the Fe(III)-EDTA complex have a high rate of absorption of iron. Ferrous sulphate as well as Fe(III)-EDTA is suitable to enrich sugar, but while iron from ferrous sulphate is precipitated and poorly absorbed when fortified sugar is added to beverages such as tea, Fe(III)-EDTA reacts slowly with tea and iron is not precipitated for at least 24 hr. Fe(III)-EDTA, as iron fortification, has demonstrated so far more advantages than that observed from other iron salts, including ferrous sulphate. But EDTA is a chelating agent and its use in food technology to prevent oxidative damage to food has been restricted. Excessive consumption of EDTA can cause abdominal cramps, nausea, low blood pressure and damage to kidneys. According to the National Institute of Health, it is unsafe to consume more than 3 g of EDTA per day or continuously for more than 5 to 7 days. The amount of EDTA necessary for 10 mg of iron fortification is about 60 mg. This is within the safe limits and is comparable to the usual amount added to the diet. |
Based on the information provided above, answer the following questions:
- Why is the Fe(III)-EDTA complex stable as compared to ferrous sulphate? (1)
OR
What happens when hard water is titrated against Na2EDTA? - You are a doctor working in Somalia. Will you recommend iron-fortified food to your patients? Support your answer with references from the passage. (1)
-
- What is the denticity of the ligand in the Fe(III)-EDTA complex? Name the atom(s) through which it can bind to the central metal ion. (1)
- Write the structure of EDTA. (1)
Chapter:
Write the structure of the expected product of the Cannizzaro reaction of 2-chlorobenzaldehyde.
Chapter:
Advertisements
How would the presence of an −SO3H group affect the basic strength of aniline?
Chapter:
Write the steps to prepare Benzoic acid from Benzoyl chloride.
Chapter:
Propanal and Propanone
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
Write the structure of the expected product of the Wolf-Kishner reduction of 2-methylbutanal.
Chapter:
How would the presence of a −SO₃H group affect the acidic strength of benzoic acid?
Chapter:
Give a simple chemical test to distinguish between the following pair of compounds:
Ethanal and Propanal
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
Identify and give any one point of difference between the protein present in the hair and protein present in egg albumin.
Chapter:
Both glucose and sucrose have an aldehydic group, then why does only glucose reduces Fehling’s Reagent and not sucrose?
Chapter:
Give a reason for the following observations:
Amino acids behave like salts.
Chapter: [10] Biomolecules
What chemical change takes place during curdling of milk?
Chapter:
Doctor advised a 50 year old woman to get enough exposure to sunlight and add fish and egg yolk to her diet. What possible disease did the doctor diagnose?
Chapter:
Identify and give any one point of difference between the carbohydrate present in cane sugar and the carbohydrate present in milk.
Chapter:
Glucose is an aldohexose and a monosaccharide. Which oxidising agent should be used to bring about oxidation of only the aldehydic group present in glucose?
Chapter:
Amino acid HOOC–CH2CH(NH2)CH2COOH. Predict whether the pH of this amino acid will be > 7, 7 or < 7.
Chapter:
Name the two major molecular shapes formed due to the folding of the secondary structure of proteins.
Chapter:
Ashish’s gums bleed frequently. The doctor’s prescription mentioned that Ashish is suffering from scurvy. Help him to identify two food sources to help him recover faster.
Chapter:
The rate of a reaction triples when the temperature changes from 298 K to 318 K. Calculate the energy of activation of the reaction, assuming that it does not change with temperature.
(Given: R = 8.314 JK−1 mol−1, log 3 = 0.4771)
Chapter:
Identify the order of reaction and write its integrated rate equation, mentioning what each term in the equation represents.
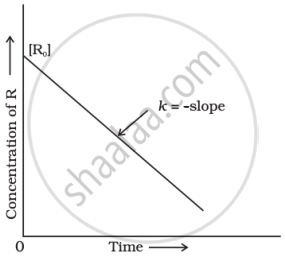
Chapter:
Consider the following first-order thermal decomposition of SO2Cl2 at a constant volume.
\[\ce{SO2Cl2_{(g)}->SO2_{(g)} + Cl2_{(g)}}\]
If the total pressure of the gases is found to be 200 torr after 10 seconds and 300 torr upon the complete decomposition of SO2Cl2. Calculate the rate constant.
(Given log 3 = 0.4771, log 2 = 0.3010)
Chapter:
For a bimolecular elementary reaction, \[\ce{A + B -> Products}\]. Write the expression for the rate of reaction relating temperature and activation energy for the reaction and also mention what each term represents in the equation.
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 12 Chemistry with solutions 2025 - 2026
Previous year Question paper for CBSE Class 12 -2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 12.
How CBSE Class 12 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.