Advertisements
Advertisements
प्रश्न
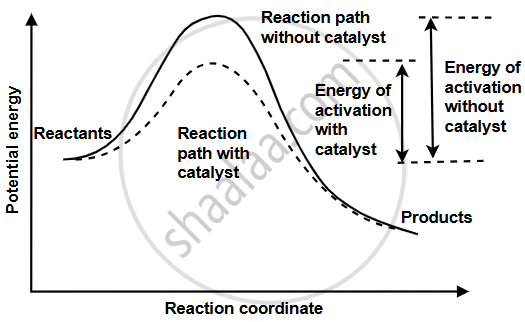
Consider the graph for the reaction:
\[\ce{H2 + I2 {⇌} 2HI}\]

- Calculate enthalpy of reaction and activation energy for the backward reaction.
- How will the catalyst affect the rate of this reaction? Explain.
स्पष्ट कीजिए
संख्यात्मक
Advertisements
उत्तर
I. Enthalpy of reaction:
`(Δ"H") = "E"_("products") - "E"_"reactants"`
= 100 − 90
= +10 kJ mol−1
Activation energy for backward reaction:
`"E"_"a"("back") = "E"_"activated complex" - "E"_"products"`
= 140 − 100
= 40 kJ mol−1
II. Catalyst will increase the rate of reaction, as the activation energy required to form an intermediate activated complex between the reactant and catalyst is lower than the activation energy required for forming a complex without a catalyst.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
