Advertisements
Advertisements
प्रश्न
Write the structural formulae for the following name and also write correct IUPAC names for them.
5-ethyl-3-methylheptane
Advertisements
उत्तर
| Given name | Structural formula | CORRECT IUPAC name |
| 5-Ethyl-3-methylheptane | 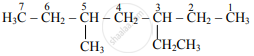 |
3-Ethyl-5-methylheptane |
APPEARS IN
संबंधित प्रश्न
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\\\phantom{...}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{...}\\\ce{H - C - C - C - C - H}\\|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\\end{array}\]
Write bond-line formulae and condensed formulae for the following compound.
3-methyloctane
Write the structural formulae for the following name and also write the correct IUPAC name for it.
2,2,3-trimethylpentan-4-ol
Distinguish between Carbocation and carbanion.
The condensed formula of the following compound is ____________.

The following structure can be represented as:
In wedge formula, the solid wedge is used to indicate ____________.
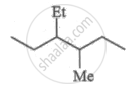
What is the molar mass of a compound represented below?
Write dash formulae for the following bond line formulae.

Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H} \phantom{...}\ce{H}\phantom{...} \ce{H} \phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...} \ce{H}\phantom{...} \ce{H}\phantom{...} \ce{H}
\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{......}\ce{H}\phantom{...} \ce{H} \phantom{...}\ce{H}\phantom{....} \ce{O}\\
\phantom{......}|\phantom{....}|\phantom{....}|\phantom{....}// \\
\ce{H - C - C - C - C }\\
\phantom{......}|\phantom{....}|\phantom{....}|\phantom{.....}\backslash \\
\phantom{..........}\ce{H}\phantom{...} \ce{H} \phantom{...}\ce{H}\phantom{....} \ce{OH}\phantom{...}
\end{array}\]
Write bond line formulae and condensed formulae for the following compound.
2, 2, 4, 4- tetramethylpentane
Write bond line formulae and condensed formulae for the following compound.
2, 2, 4, 4- tetramethylpentane
Write the structural formulae for the following names and also write correct IUPAC names for them.
2,2,3-trimethylpentan-4-ol
Write dash formula for the following bond line formula.
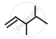
Write dash formulae for the following bond line formulae.

Write the structural formulae for the following name and also write the correct IUPAC name for them.
2,2,3-trimethylpentan-4-0l
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{N ≡ C - C - C - C ≡ N}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\\
\end{array}\]
Write condensed formulae and bond line formulae for the following structures.
\[\begin{array}{cc}
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.}\\
\phantom{.}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.}\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.}\\
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\
\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H\phantom{...}H}\\
|\phantom{....}|\\
\ce{N ≡ C - C - C - C ≡ N}\\
|\phantom{....}|\\
\ce{H\phantom{...}H}
\end{array}\]
Write dash formulae for the following bond line formulae.
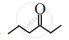
Write dash formulae for the following bond line formulae.

Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\\\phantom{...}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{...}\\\ce{H - C - C - C - C - H}\\|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}
\end{array}\]
Write condensed formulae and bond line formula for the following structure.
\[\begin{array}{cc}
\phantom{.......}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.....}\ce{O}\\
\phantom{.......}|\phantom{....}|\phantom{....}|\phantom{...} \phantom{..}//\\
\ce{H - C - C - C - C}\\
\phantom{......}|\phantom{....}|\phantom{....}|\phantom{.....}\backslash\\
\phantom{.........}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{......}\ce{OH}\\
\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{N ≡ C - C - C - C ≡ N}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}
\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{.........}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\ce{O}\\
\phantom{........}|\phantom{....}|\phantom{....}|\phantom{......}//\\
\ce{H - C - C - C - C}\\
\phantom{.......}|\phantom{....}|\phantom{....}|\phantom{......}\backslash\\\
\phantom{..........}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\ce{OH}\\
\end{array}\]
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{N ≡ C - C - C - C ≡ N}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}
\end{array}\]
Which from following methods of three-dimensional representation of molecules uses a long slanting line for C – C single bond?
