Advertisements
Advertisements
प्रश्न
Write the equation for the preparation of 1-iodobutane from 1-chlorobutane.
How would you prepare 1-iodobutane from 1-chlorobutane? Mention the equations only.
Advertisements
उत्तर
\[\ce{CH3 - (CH2)2 - CH2 - Cl + NaI ->[dry acetone][Finkelstein reaction]CH3 - (CH2)2 - CH2I + NaCl}\]
APPEARS IN
संबंधित प्रश्न
Why is sulphuric acid not used during the reaction of alcohols with KI?
A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
Write the equation for the preparation of 1-iodobutane from 1-butanol.
How will you bring about the following conversion?
Propene to 1-nitropropane
How will you bring about the following conversion?
Propene to propyne
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2Cl + NaI ->[acetone][heat]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH = CH2 + HBr->[peroxide]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH = C(CH3)2 + HBr ->}\]
How the following conversion can be carried out?
Propene to propan-1-ol
How the following conversion can be carried out?
2-Methyl-1-propene to 2-chloro-2-methylpropane
How the following conversion can be carried out?
But-1-ene to n-butyliodide
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields four isomeric monochlorides.
3-Methyl-pent-2-ene of peroxide forms an addition product. The number of possible stereoisomers for the product is ______.
Predict the major product ‘P’ in the following sequence of reactions:
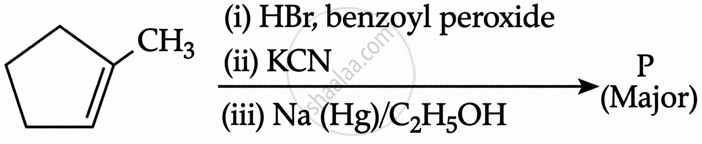
How many products (including stereoisomers) are expected from the monochlorination of the following compound?
\[\begin{array}{cc}
\ce{H3C}\phantom{..................................}\\
\backslash\phantom{...........................}\\
\ce{CH - CH2 - CH3}\\
/\phantom{...........................}\\
\ce{H3C}\phantom{...................................}
\end{array}\]
