Advertisements
Advertisements
प्रश्न
Two liquids (A) and (B) can be separated by the method of fractional distillation. The boiling point of liquid (A) is less than boiling point of liquid (B). Which of the liquids do you expect to come out first in the distillate? Explain.
Advertisements
उत्तर
When the difference in boiling points of two liquids is not much then simple distillation cannot be used to separate them. Vapours of such liquids are formed within the same temperature range and are condensed simultaneously. The technique of fractional distillation is used in such cases. In this technique, vapours of a liquid mixture are passed through a fractionating column before condensation. The fractionating column is fitted over the mouth of the round bottom flask. The liquid (A) with low boiling point will distill first.
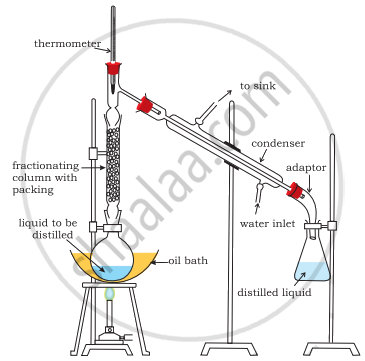
APPEARS IN
संबंधित प्रश्न
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than 25°C), which are miscible with each other?
What is the difference between distillation, distillation under reduced pressure and steam distillation ?
Define :- Distillation
How is distillation more advantageous than evaporation?
On what principle are the following methods of separation based? Give one example of a mixture for each of the methods mentioned in which they are used
Fractional distillation :
Give one word answer
The liquid which evaporates and then condenses during the process of distillation
Give one word answer
The process by which two miscible liquids are separated
Select the correct alternative from the choices given for the following statement:
Components of crude petroleum can be separated by
Give a scientific reason.
A wet cloth is wrapped around a water storage container in summer.
Write an answer to the following question in your own words.
Where are the methods of separation by distillation and by separating funnel used? Why?
State the correct technique for the separation of the following mixture.
a low boiling point liquid from a high boiling point liquid.
What will happen if the upper outlet of the condenser is connected to the tap instead of the lower outlet?
What is the difference between simple distillation and fractional distillation?
Which of the tubes in Fig. 2.1 (a) and (b) will be more effective as a condenser in the distillation apparatus?

Assertion (A): Simple distillation can help in separating a mixture of propan-1-ol (boiling point 97°C) and propanone (boiling point 56°C).
Reason (R): Liquids with a difference of more than 20°C in their boiling points can be separated by simple distillation.
Draw a diagram of bubble plate type fractionating column. When do we require such type of a column for separating two liquids. Explain the principle involved in the separation of components of a mixture of liquids by using fractionating column. What industrial applications does this process have?
Explain the factors, which will affect the rate of evaporation, taking examples from our daily life experiences.
