Advertisements
Advertisements
प्रश्न
The reagents used to convert phenol to 2,4,6-tribromophenol is _____________
विकल्प
Br2/CS2
Br2/CCl4
Br2/H2O
KBr aq.
Advertisements
उत्तर
The reagents used to convert phenol to 2,4,6-tribromophenol is Br2/H2O
संबंधित प्रश्न
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Answer in one sentence/ word.
Write the name of the electrophile used in Kolbe’s Reaction.
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
Reaction between Grignard reagent and aldehyde other than formaldehyde leads to formation of _______________
Draw intramolecular hydrogen bonding structure in o-nitrophenol.
What is the action of following reagents on pent-3-enal?
- H2/Ni
- LiAlH4/H3O+
Which of the following reagents are required in the conversion of phenol to salicylic acid?
Which of the following compounds does not react with bromine in alkaline medium?
Which of the following compounds is obtained, when phenol react with bromine water?
In phenols, −OH group is attached to ___________ hybridised carbon.
Propane when treated with cold cone. H2SO4 forms a compound which on heating with water gives ______.
The number of isomeric alcohols possible with the formula C4H10O is ____________.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
____________ will NOT undergo acetylation.

Identify reagents X, Y and Z.
Which of the following reagents is used to convert phenol to benzene?
\[\ce{Isopropyl alcohol + acidic K2Cr2O7 -> X}\]
Identify product 'X' in the above reactions.
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
Which isomer of C4H10O is optically active?
Identify the product X in the following reaction.
\[\ce{Phenol ->[Na2Cr2O7][H2SO4] X}\]
Which among the following phenolic compound is most acidic in nature?
Name the catalyst used in commercial method of preparation of phenol.
The acid, which contains both -OH and -COOR groups is ______.
Which of the following compounds has lowest boiling point?
The major product obtained in the following reaction is

Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Arrange the following compounds in order of decreasing acidity:
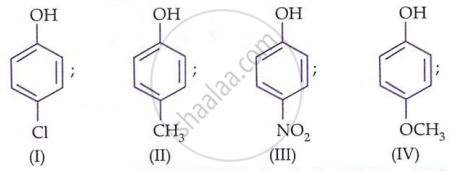
Which of the following is trihydric phenol?
