Advertisements
Advertisements
प्रश्न
The electronic structures of atoms P and Q are shown below:
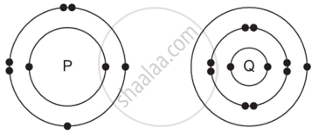
Based on the information given above, answer the following questions:
- If P and Q combine to form a compound, what type of bond is formed between them?
- Give the chemical formula of the compound formed.
- The compound so formed is dissolved in water. Is the resultant solution acidic or basic in nature? Justify your answer.
- Write the chemical equation for the reaction between ‘Q’ and ethanol.
- What will be the formula of the compound formed when ‘P’ undergoes bonding with carbon?
रासायनिक समीकरण/संरचनाएँ
औचित्य
लघु उत्तरीय
Advertisements
उत्तर
- The bond formed after the combination of P and Q is an ionic bond.
- The chemical formula of the compound formed is Q2P.
- Basic, metallic oxides are basic in nature.
- The chemical equation is as follows:
\[\ce{2C2H5OH + 2Q -> 2C2H5OQ + H2}\] - The formula of the compound formed when ‘P’ undergoes bonding with carbon is CP2.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
