Advertisements
Advertisements
प्रश्न
A hydrocarbon with the formula CxHy undergoes complete combustion as shown in the following equation:
\[\ce{2C_xH_y + 9O2 -> 6CO2 + 6H2O}\]
- What are the values of ‘x’ and ‘y’?
- Give the chemical (IUPAC) name of the hydrocarbon.
- Draw its electron dot structure.
- Name the alcohol which on heating with conc. H2SO4 will produce the above hydrocarbon CxHy.
- Write a balanced chemical equation for the reaction of CxHy with hydrogen gas in presence of Nickel.
रासायनिक समीकरण/संरचनाएँ
लघु उत्तरीय
Advertisements
उत्तर
a. The values of ‘x’ and ‘y’ are x = 3, y = 6.
b. The IUPAC name of the hydrocarbon is propene.
c. The electron dot structure is as follows:
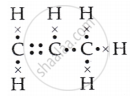
d. The name of the alcohol is propanol.
e. The balanced chemical equation is:
\[\ce{C3H6 + H2 ->[Ni] C3H8}\]
\[\ce{CH2 = CH - CH3 + H2 ->[Ni] CH3 - CH2 - CH3}\]
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
