Advertisements
Advertisements
प्रश्न
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
Advertisements
उत्तर
(i) Ammonia burns with a yellowish flame. It produces water vapour and nitrogen.
(ii) When ammonium hydroxide is brought near the mouth of concentrated hydrochloric acid, it produces dense white fumes of ammonium chloride.
APPEARS IN
संबंधित प्रश्न
What property of ammonia is demonstrated by the fountain experiment?
Name two gases which give dense white fumes with ammonia.
Choose the correct word or phrase from the bracket to complete the following sentence:
Heating ammonium chloride with sodium hydroxide produces ______.
State one relevant observation of burning of ammonia in air.
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
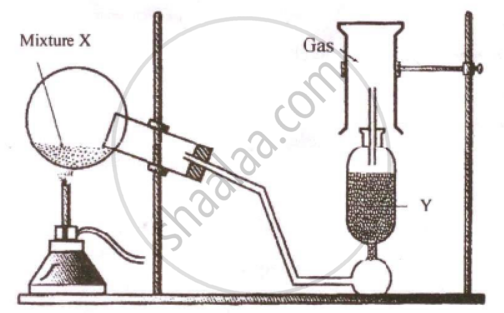
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Ammonia in excess is mixed with chlorine.
Differentiate between:
Action on indicators or dry ammonia gas and aqueous ammonia
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
