Advertisements
Advertisements
प्रश्न
Name the elements in Period 1.
Advertisements
उत्तर
Names of elements are (H) Hydrogen and Helium (He).
APPEARS IN
संबंधित प्रश्न
The elements 4Be, 12Mg and 20Ca, each having two valence electrons in their valence shells, are in periods 2, 3 and 4 respectively of the modern periodic table. Answer the following questions associated with these elements, giving reason in each case:
(a) In which group should they be?
(b) Which one of them is least reactive?
(c) Which one of them has the largest atomic size?
Fill in the blank in the following statement:
On going down in a group in the periodic table, the metallic character of elements ...............
How does the electropositive character of elements change on going down in a group of the periodic table?
How do atomic structures (electron arrangements) change in a period with increase in atomic numbers from left to right?
Explain why, the properties of elements are repeated after 2, 8, 18 and 32 elements in the periodic table.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 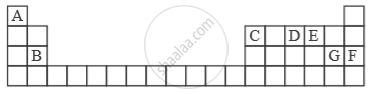
The ion of which element will migrate towards the cathode during electrolysis?
Choose the most appropriate answer from the following list of oxides which fit the description.
An oxide which dissolves in water forming an acid.
Choose the most appropriate answer from the following list of oxides which fit the description.
An amphoteric oxide: Al2O3 (shows both acidic and basic properties)
Write the electronic configuration of element 17T35.
- What is the group number of T?
- What is the period number of T?
- How many valence electrons are there in an atom of T?
- What is the valency of T?
- Is it a metal or non-metal?
- State number of protons and neutrons in T.
Name and state the following with reference to the elements of the first three periods of the periodic table.
Valency of elements in Group 14 and 15.
