Advertisements
Advertisements
प्रश्न
Name one solid, one liquid and one gaseous non-metal.
Advertisements
उत्तर
Carbon is a solid non-metal, bromine is a liquid non-metal and oxygen is a gaseous non-metal.
APPEARS IN
संबंधित प्रश्न
State two characteristics of matter demonstrated by Brownian motion.
What is Brownian motion? Draw a diagram to show the movement of a particle (like a pollen grain during Brownian motion.
In a beam of sunlight entering a room, we can sometimes see dust particles moving in a haphazard way in the air. Why do these dust particles move?
What elements do the following compounds contain ?
Sugar, Common salt
Which of the following are ‘pure substances’ ?
Ice, Milk, Iron, Hydrochloric acid, Calcium oxide, Mercury, Brick, Wood, Air
Give reason why graphite is used for making elecrode in a dry cell.
What are (i) metals (ii) non-metals, and (iii) metalloids ? Give two examples each of metals, non-metals and metalloids.
The element which is not common between the compounds called baking soda and soda ash is
In the following set of substances, one item does not belong to the set. Select this item and explain why it does not belong to the set :
Hydrogen, Oxygen, Steam, Chlorine
Out of a colloid, solution and a suspension :
which one has the largest particles ?
How much water should be added to 15 grams of salt to obtain 15 per cent salt solution ?
Choose one term from the following which includes the other three :
aerosol, emulsion, colloid, sol
Which of the following is a sol ?
Shaving cream, Milk, Fog, Soap solution, Hairspray
You have been given a suspension and a solution. How could you tell the difference between them by their appearance ?
What type of magnet is fitted on a crane to separate scrap iron objects from a heap of waste materials in factories ?
Asmita went to buy an earthen pot. There she saw many things kept for sale.
What is the difference between a substance and an object?
Justify your answer.
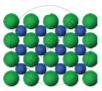
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Name the metals that are used in jewellery.
The non S I and S I units of some physical quantities are given in column A and column B respectively. Match the units belonging to the same physical quantity:
| (A) | (B) | ||
| (a) | degree celsius | (i) | kilogram |
| (b) | centimetre | (ii) | pascal |
| (c) | gram per centimetre cube | (iii) | metre |
| (d) | bar | (iv) | kelvin |
| (e) | milligram | (v) | kilogram per metre cube |
Solids possess very high kinetic energy.
