Advertisements
Advertisements
प्रश्न
Name the gas evolved when dilute hydrochloric acid is added to: Sodium sulphite
Advertisements
उत्तर
Sulphur dioxide
APPEARS IN
संबंधित प्रश्न
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Complete the following reaction and balance them.
Ca(HCO3)2 + 2HCl →
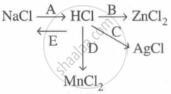
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Calcium oxide and phosphorous pentoxide are very good drying agents but they are not used to dry HCI gas. Give reasons for each.
Name the gas evolved when dilute hydrochloric acid is added to: Lead (II) sulphide
Name the gas evolved when dilute hydrochloric acid is added to: Potassium bisulphite
How will you identify?
Chlorine gas
- Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen chloride
- Chlorine
- Carbon dioxide
- Oxygen
- Hydrogen
- Select a basic gas mentioned in Q. (a). How is the basic nature suspected?
- Select acidic gases from the gases mentioned in Q. (a). How is the acidic nature suspected?
- The two gases A and B are bleaching agents. A is greenish-yellow and bleaches due to its oxidizing property while B is a colourless gas that bleaches due to reduction. Identify A and B?
- Which gas turn blue cobalt chloride paper light pink? Give one similarity in the test between
- Cl2 and HCl
- SO2 and CO2.
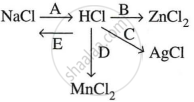
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
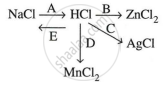
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.