Advertisements
Advertisements
प्रश्न
Name the gas evolved when dilute hydrochloric acid is added to: Calcium carbonate
Advertisements
उत्तर
Carbon dioxide
APPEARS IN
संबंधित प्रश्न
Name a chloride which is solube in excess of ammonium hydroxide
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb3O4 + HCl ->}\]
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
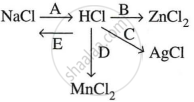
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Give a balanced equation when dilute hydrochloric acid is added to : Magnesium bicarbonate
How will you identify?
Chlorine gas
Complete and balance the following reaction, state whether dilutes or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Complete and balance the following reaction; state whether it is dilute or cone. acid is used.
\[\ce{NH_4OH + HCl ->}\]
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.