Advertisements
Advertisements
प्रश्न
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Advertisements
उत्तर
\[\ce{NH4OH + HCl (dil{.}) -> NH4Cl + H2O}\]
Dilute HCl is used.
APPEARS IN
संबंधित प्रश्न
Name two gases which chemically combine to form a liquid.
Name the gas evolved when dilute hydrochloric acid is added to: Calcium carbonate
Name the gas evolved when dilute hydrochloric acid is added to: Magnesium bicarbonate
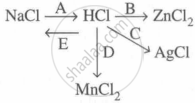
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.

Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
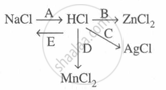
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
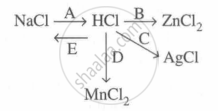
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.