Advertisements
Advertisements
प्रश्न
Moving left to right in the second period, the number of valence electrons _____.
Advertisements
उत्तर
Moving left to right in the second period, the number of valence electrons increases from 1 to 8.
APPEARS IN
संबंधित प्रश्न
Based on the group valency of elements state the formula lot the following giving justification for each :-
(i) Oxides of 1st group elements,
(ii) Halides of the elements of group 13, and
(iii) Compounds formed when an element of group 2 combines with, an element of group 16.
Calculate the mass of Calcium that will contain the same number of the atom as are present in 3.2 gm of Sulphur.
[Atomic masses: S=32, Ca=40]
How does the tendency to lose electrons change as we go down in group 1 of the periodic table? Why does it change this way?
Write two reasons responsible for late discovery of noble gases ?
K, Pb, Ca, Zn (In the increasing order of the reactivity)
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 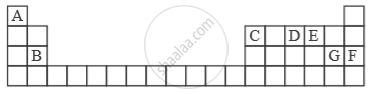
The ion of which element will migrate towards the cathode during electrolysis?
Select the correct answer
Identify the metalloid
Atomic number of an element is 16. State
- the period to which it belongs
- the number of valence electrons
- whether it is a metal or non-metal
Atomic numbers of elements A, B, C, D, E, F are 8, 7, 11, 12, 13 and 9 respectively. State the type of ions they form.
