Advertisements
Advertisements
प्रश्न
How will you convert nitromethane into dimethylamine?
Advertisements
उत्तर
\[\ce{\underset{Nitromethane}{CH3NO2} ->[Sn/HCl] \underset{Methyl amine}{CH3NH2} ->[CHCl3 + KOH] CH3NC ->[Na/C2H5OH][(Reduction)] \underset{Dimethylamine}{CH3NHCH3}}\]
APPEARS IN
संबंधित प्रश्न
Which among the following molecular formulae represents urotropine?
(a) C6H12N4
(b) C6H24H4
(c) C6H12N4O2
(d) C6H24N4O2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
C6H5NHCH3
Give one chemical test to distinguish between the following pair of compounds.
Methylamine and dimethylamine
Give one chemical test to distinguish between the following pair of compounds.
Ethylamine and aniline
How will you convert hexanenitrile into 1-aminopentane?
How will you convert methanol to ethanoic acid?
How will you convert propanoic acid into ethanoic acid?
Complete the following reaction:
\[\ce{C6H5NH2 + H2SO4 (conc.)}\]
Complete the following reaction:
\[\ce{C6H5N2Cl ->[(i) HBF4][(ii) NaNO2/Cu, \Delta]}\]
Give reasons Although –NH2 is o/p directing group, yet aniline on nitration gives a significant amount of m-nitroaniline
Do the following conversions in not more than two steps :
Ethyl benzene to Benzoic acid
Do the following conversions in not more than two steps :
Propanone to Propene
Write the structure of 2,4-dinitrochlorobenzene
What is the action of p-toluenesulphonychloride on ethylamine and diethylamine?
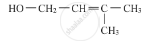
Write the IUPAC name of the given compound :
Using IUPAC norms, write the formula of Hexaamminecobalt(III) sulphate.
Do the following conversions in not more than two steps:

