Advertisements
Advertisements
प्रश्न
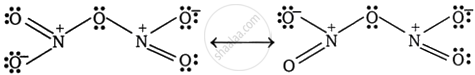
Give the resonating structure of N2O5.
Advertisements
उत्तर
संबंधित प्रश्न
What is the covalence of nitrogen in N2O5?
Bond angle in `PH_(4)^(+)` is higher than that in PH3. Why?
How is nitrogen prepared in the laboratory? Write the chemical equations of the reactions involved.
Explain why NH3 is basic while BiH3 is only feebly basic.
Which of the following is a basic oxide?
(A) SiO2
(B) P4O10
(C) MgO
(D) Al2O3
Explain why Mn2+ ion is more stable than Mn3+? (Given : Mn→ Z=25)
The statement true for \[\ce{N^-_3}\] is ____________.
Nitrogen is relatively inactive element because ____________.
Among the 15th group elements, as we move from nitrogen to bismuth, the pentavalency becomes less pronounced and trivalency becomes more pronounced due to:
Pure nitrogen is prepared in the laboratory by heating a mixture of:
The bonds present in N2O5 are ____________.
Which of the following oxides of nitrogen reacts with FeSO4 to form a dark brown compound?
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
A deep brown gas is formed by mixing two colourless gases which are:
The laughing gas is ____________.
Name three oxoacids of nitrogen. Write the disproportionation reaction of that oxoacid of nitrogen in which nitrogen is in +3 oxidation state.
On heating lead (II) nitrate gives a brown gas “A”. The gas “A” on cooling changes to colourless solid “B”. Solid “B” on heating with NO changes to a blue solid ‘C’. Identify ‘A’, ‘B’ and ‘C’ and also write reactions involved and draw the structures of ‘B’ and ‘C’.
HCl is added to following oxides. Which one would give H2O2
Heating white phosphorus with conc. NaOH solution gives mainly ______.
