Advertisements
Advertisements
प्रश्न
Give reason:
Reactions involving Grignard reagent must be carried out under anhydrous condition.
Advertisements
उत्तर
Grignard reagents are highly reactive compounds. They react with water or compounds containing hydrogen attached to the electronegative element. Hence, reactions involving the Grignard reagent must be carried out under anhydrous condition.
संबंधित प्रश्न
Explain reactions of haloarenes with sodium metal.
The most suitable reagent(s) required to prepare 1-iodobutane from but-1-ene is/are ____________.
Identify the compound that gives 2, 5-dimethylhexane by reacting with sodium metal in presence of dry ether.
What is the action of following on ethyl bromide:
Na in dry ether
What is the action of following on ethyl bromide?
\[\ce{Mg}\] in dry ether
Write chemical reactions for the following conversions:
Bromobenzene to toluene.
The molecular formula of Wilkinson's catalyst used in the hydrogenation of alkenes is ______.
Nitration ofwhich among the following compounds yields cyclonite?
The Wurtz-Fittig reaction involves condensation of ______.
Identify the product obtained in the following reaction.
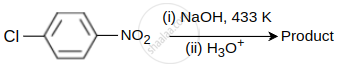
The major product obtained in the following reaction is
\[\ce{Chlorobenzene + Chlorine ->[Anhydrous][FeCl3] product (Major)}\]
Which of the following alkyl halide is treated with sodium metal to obtain 2, 2, 3, 3 tetramethyl butane?
Write the structure of the product formed when chlorobenzene is treated with sodium metal in the presence of dry ether.
Give the following named reaction.
Fittig reaction
Write the product formed during Chlorobenzene treated with chlorine reactions.
Write the product formed by chlorobenzene treated with a fuming H2SO4 reaction.
Write the product formed Anisole treated with HI reaction.
Define organometallic compound.
What is the action of following on ethyl bromide?
silver nitrite
Identify the product 'Z' in the following series of reactions.
\[\ce{\underset{}{Ethanol} ->[SOCl2][Δ] \underset{\underset{}{}}{X}->[Mg][Dry ether] \underset{\underset{}{}}{Y}->[NH3] \underset{\underset{}{}}{Z}}\]
