Advertisements
Advertisements
प्रश्न
Give reason:
Alkyl halides are generally not prepared by free radical halogenation of alkanes.
Advertisements
उत्तर
Free radical halogenation of alkanes leads to the formation of a mixture of mono and poly halogen compounds. Hence, free radical halogenation of alkanes is not suitable for the preparation of alkyl halides.
संबंधित प्रश्न
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH2Cl + NaI ->[Acetone]}\\
|\phantom{..................}\\
\ce{CH3\phantom{...............}}
\end{array}\]
Write the structure and IUPAC name of the major product in the following reaction.
\[\ce{CH3 - CH2Br + SbF3 ->}\]
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH = CH2 + HBr ->[peroxide]}\\
|\phantom{........................}\\
\ce{CH3\phantom{.....................}}
\end{array}\]
Write the structure and IUPAC name of the major product in the following reaction.

Convert the following:
But-1-ene to n-butyl iodide
Aromatic electrophilic substitution with iodine can be carried out using _____________
Predict all the alkenes that would be formed by dehydrohalogenation of the following alkyl halide.
- 2-chloro-2-methylbutane
- 3-bromo-2,2,3-trimethylpentane
Which of the following pairs of aryl halides cannot be prepared directly by electrophilic substitution?
Which among the following methods is not suitable for the preparation of alkyl chlorides?
What is the name of reaction involving replacement of diazonium group by chloride using cuprous (I) salt?
Alkyl halides can be hydrolysed to alcohols by reacting with ____________.
Alkyl fluorides are prepared by heating alkyl bromides or chlorides in the presence of metallic fluorides. The reaction is known as ____________ reaction.
Conversion of an alcohol into alkyl chloride by reacting with thionyl chloride is done in a medium of ____________.
Which of the following alkyl halides has the lowest boiling point?
Which reaction is useful in exchange of halogen in alkyl chloride by iodide?
Identify the INCORRECT statement.
The reaction \[\ce{2R - Cl + COF2 -> 2R - F + COCl2}\] is an example of ______.
What is molecular formula of allyl chloride?
An alkyl iodide on refluxing with aqueous KOH solution gave isopropyl alcohol. The structure of alkyl iodide could be:
Identify the name of reaction in which alkyl fluorides are prepared by heating alkyl bromide with metallic fluorides.
Alkyl chloride when treated with sodium iodide in presence of dry acetone forms alkyl iodide. What is the name of this reaction?
The conversion of ethyl bromide using sodium iodide and dry acetone, this reaction is know as ______.
Explain Swartz reaction.
What is the advantage of alkyl halide using the thionyl chloride (SOCl2 ) method?
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}
\end{array}\]
The given compound
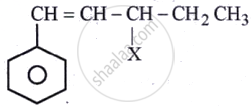
is an example of ______.
