Advertisements
Advertisements
प्रश्न
Explain the difference between Metals and non-metals.
Advertisements
उत्तर
| S. No. | Physical properties | Metals | Non-metals |
| 1. | Malleability and ductility | They can be beaten into sheets and wires. | They cannot form sheets or wires. |
| 2. | Sonority | Metals are sonorous; they produce a ringing sound when beating. | They are non−sonorous. |
| 3. | Conduction | They conduct heat and electricity. | Non-metals do not allow heat and electricity to pass through them. |
| 4. | Chemical Properties | Metals generally react by losing electrons, forming basic oxides, and conducting electricity and heat. | Non-metals usually gain or share electrons, form acidic or neutral oxides, and are poor conductors of electricity and heat. |
| 5. | Nature of oxides | Metallic oxides are basic. | Oxides of non−metals are acidic. |
| 6. | Reaction with water | Metals react with water. | Non−metals do not react with water. |
| 7. | Reaction with acids | Metals react with acids to produce hydrogen gas. | They generally do not react with acids. |
संबंधित प्रश्न
What is the difference between a pure substance and a mixture ?
Give reason: Argon is filled in electric bulbs.
Fill in the blank
A liquid non-metal is _______.
The important physical properties of substances are colour, odour, nature, density & solubility in water.
Name – two coloured gases (with their colours)
Complete the statement given below by filling in the blank with the correct word.
From the elements nitrogen, chlorine, bromine, the element present in the atmosphere is ________.
Write the names of elements from the following symbol:
Si
The diagram represents fractional distillation for the separation of mixtures. Answer the following :
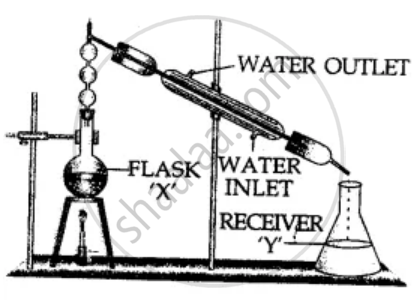
- Can two immiscible liquids be separated by this process?
- Separation of liquids by this process is based on which physical property?
- If methyl alcohol & water are to be separated, which liquid would remain in flask ‘X’ after condensation.
- Give a reason for the above answer.
- State the purpose of the fractionating column in the apparatus.
Classify the following as elements and compounds.
Water, Common salt, Sugar, Carbon dioxide, Iodine and Lithium
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
Name a non-metal which is required for combustion.
What is the Latin name of Sodium?
