Advertisements
Advertisements
प्रश्न
Explain concept with example/explain with the help of a balanced equation.
Gangue
Advertisements
उत्तर
- The unwanted impurities of sand, soil, rocky substances, etc., present in the ore are called gangue.
- Ores are taken out from the mines and the gangue is usually separated from the ore at the site itself by various methods.
- The process of separating gangue from the ores is called the concentration of ores.
- For example, bauxite ore contains impurities of silica (SiO2), ferric oxide (Fe2O3) and titanium oxide (TiO2). These impurities are called gangue.
APPEARS IN
संबंधित प्रश्न
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Identify A and B and give their reactions with dil HCL and dil H2SO4 ?
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their ?
oxides
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: nitrates
Explain the following term:
Gangue
Explain the following terms:
(c) slag
Explain the following terms:
smelting
What do you observe when hydrogen is passed over heated copper oxide?
`Ag2O` ................................
................................
`ZnCO_3`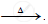 ..............................
..............................
`2Pb(NO3)_2`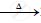 .........................
.........................
Distinguish between electrolytic methods of reduction and refining.
Arrange the metals in (2) in the decreasing order of reactivity.
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
A compound that is added to lower the fusion temperature of the electrolytic bath in the extraction of aluminium.
Match the columns.
| Group A | Group B |
| 1. ZnS | a) Copper Sulphide |
| 2. HgS | b) Bauxite |
| c) Cinnabar | |
| d) Zinc blend |
Cassiterite is a copper ore.
Write the molecular formulae of the following compound.
Ferrous tungstate
Distinguish between:
Froth floatation - Leaching
The chemical name of rust is ______.
Which of the following methods is used to separate wolframite and stannic oxide present in cassiterite?
