Advertisements
Advertisements
प्रश्न
Distinguish between the following:
Hypotonic and Hypertonic solutions
Advertisements
उत्तर
| Hypotonic solutions | Hypertonic solutions | |
| (i) | In this condition, the solution outside the cell has a lower solute concentration than the cell sap. | In this condition, the solution outside the cell has a higher solute concentration than the cells sap. |
| (ii) | The water molecules from outside will move into the cell (endosmosis). | The water molecules from the interior of the cell will move out (exosmosis) under excessive root pressure. |
| (iii) | It is a weak solution and strong solvent. | It is a strong solution with a weak solvent. |
संबंधित प्रश्न
A candidate in order to study the process of osmosis has taken 3 potato cubes and put them in 3 different beakers containing 3 different solutions. After 24 hours, in the first beaker the potato cube increased in size, in the second beaker the potato cube decreased in size and in the third beaker there was no change in the size of the potato cube. The following diagram shows the result of the same experiment:

(i) Give the technical terms of the solutions used in beakers, 1, 2 and 3.
(ii) In beaker 3, the size of the potato cube remains the same. Explain the reason in brief.
(iii) Write the specific feature of the cell sap of root hairs which helps in absorption of water.
(iv) What is osmosis?
(v) How does a cell wall and a cell membrane differ in their permeability?
Movement of molecules of a substance from the region of their higher concentration to the region of their lower concentration without the involvement a separating membrane, is called
The most appropriate characteristic of a semipermeable membrane is that
Name the following:
The condition of a cell placed in a hypotonic solution.
The diagram given below represents an experimental set-up to demonstrate a certain process. Study the same and answer the questions that follow:
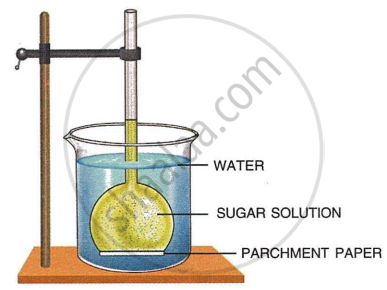
- Name the process.
- Define the above-named process.
- What would you observe in the experimental set-up after an hour or so?
- What control experiment can be set up for comparison?
- Keeping in mind the root hair, cell and its surroundings, name the parts that correspond to
- concentrated sugar solution
- parchment paper
- water in the beaker
- Name any other material that can be used instead of parchment paper in the above experiment.
- Mention two advantages of the process for the plants.
Show by a series of diagrams, the change which a plant cell will undergo when placed in
Hypertonic salt solution
Define the following:
Diffusion
Name the following:
Movement of molecules from a region of high concentration to a region of low concentration.
Choose the correct answer:
Plasmolysis will occur when the cell is placed in a __________
A leaf cell of a water plant was placed in a liquid other than pond water. After sometime, it assumed a shape as shown below:

Comment on the nature (tonicity) of the liquid surrounding the cell.
What do you mean by transpiration pull ?
Differentiate between the following
Endosmosis and Exosmosis
Give a diagrammatic representation of plasmolysis in a cell.
The diagram given below is of an experiment just at the start. Study the diagram carefully and answer the following questions :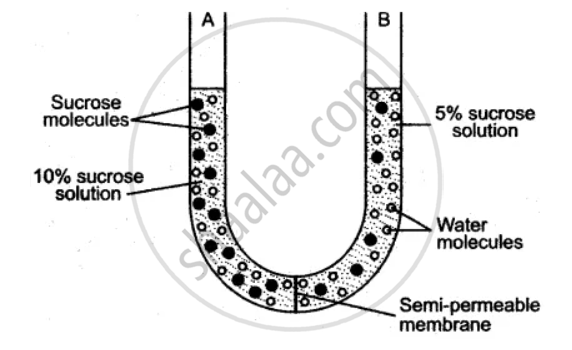
(i) What does the experiment demonstrate?
(ii) Define, the process demonstrated in the experiment.
(iii) What changes are observed after a few hours?
(iv) Give two examples of a semi-permeable membrane.
(v) Which limb of the U-tube contains a more concentrated sucrose solution, A or B?
(vi) Why is the membrane separating the two solutions labeled as semi-permeable membrane?
A plant cell kept in a drop of water was examined under the low power magnification of a microscope, as shown:
(i) What would you do to bring this cell back to its original condition?
(ii) What scientific term is used for such condition?
(iii) Draw the same cell if it is kept in a strong sugar solution.
Mention, if the following statement is True or False. If false rewrite the wrong statement in its correct form:
During exosmosis, water moves from inside the cell to the outside of the cell.
Multiple Choice Question:
Marine fish when thrown under tap water bursts because of:
Name the two organelles in a plant cell that contain their own genetic material and ribosomes.
During a practical exam, a plant cell in a particular solution was placed under a compound microscope. Students were told to observe the cell and name the tonicity of the solution and mention the process that occurred in the cell.

