Advertisements
Advertisements
प्रश्न
Define order of reaction. How does order of a reaction differ from molecularity for a complex reaction?
Advertisements
उत्तर
Order of Reaction :
Rate = K[A]x [B]y
(x + y) = order of that chemical reaction.
The sum of powers of the concentration of the reactants in the rate law expression is called the order of that chemical reaction.
| Order of Reaction | Molecularity |
| Sum of power of the concentration of the reactants in the rate law expression | The number of reacting species (atoms, ions or molecules) taking part in an elementing reaction, which must colloide simultaneously in order to being about a chemical reaction. |
| Order is an experimental quantity It can be 0 or fraction. |
It cannot be zero or a non- integer |
| Order is applicable to elementary as well as complex reaction |
For complex reaction, molecularity has no meaning. |
APPEARS IN
संबंधित प्रश्न
Following data are obtained for reaction :
N2O5 → 2NO2 + 1/2O2
| t/s | 0 | 300 | 600 |
| [N2O5]/mol L–1 | 1.6 × 10-2 | 0.8 × 10–2 | 0.4 × 10–2 |
1) Show that it follows first order reaction.
2) Calculate the half-life.
(Given log 2 = 0.3010, log 4 = 0.6021)
A first order reaction takes 20 minutes for 25% decomposition. Calculate the time when 75% of the reaction will be completed.
(Given : log = 2 = 0·3010, log 3 = 0·4771, log 4 = 0·6021)
A first order reaction is 50% complete in 20 minute What is rate constant?
In the first order reaction, half of the reaction is complete in 100 seconds. The time for 99% of the reaction to occurs will be
The reaction X → product
Follow first order of kinetics. In 40 minutes the concentration of 'X' changes from 0.1 m to 0.025. M. The rate of reaction when concentration of X is 0.01 m is.
Observe the graph shown in figure and answer the following questions:
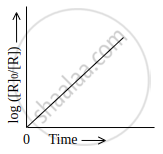
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
The slope in the plot of ln[R] vs. time for a first order reaction is ______.
Define first-order reaction.
The rate constant for the reaction:
\[\ce{2N2O_{(s)} ->2N2O4_{(g)}}\] is 4.98 × 10-4 s-1.
The order of the reaction is ______.
If the half-life (t1/2) for a first order reaction is 1 minute, then the time required for 99.9% completion of the reaction is closest to ______.
