Advertisements
Advertisements
प्रश्न
Compare the chemistry of actinoids with that of the lanthanoids with special reference to chemical reactivity.
Advertisements
उत्तर १
Generally, the initial members of the series are very reactive, like calcium, in their chemical behaviour, but as the atomic number increases, they behave like aluminium.
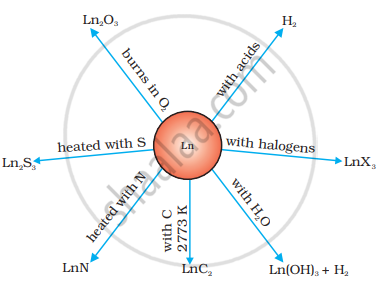
The value of E° for the half-reaction \[\ce{Ln^{3+}_{ (aq)} + 3e- -> Ln_{(s)}}\] is in the range of −2.2 V to −2.4 V. For Eu, the value of E° is −2.0 V. Of course, there is a slight variation in the value. These metals react with hydrogen when gently heated in a hydrogen atmosphere. On heating these metals with carbon, the carbides Ln3C, Ln2C3 and LnC2 are formed. These metals liberate hydrogen gas from dilute acids and, when burned in a halogen atmosphere, form halides. These form oxide M2O3 and hydroxide M(OH)3. Hydroxides are definite compounds and not just hydrated oxides. They are basic, like the oxides and hydroxides of alkaline earth metals. Their general reactions are shown in the figure.
Actinoids are very reactive metals, especially when they are finely divided. The action of boiling water on them gives a mixture of oxide and hydride, and combines with most non-metals at normal temperatures. Hydrochloric acid affects all metals, but most metals are little affected by nitric acid. The reason is that a protective oxide surface forms on these metals. Alkalis have no effect on these metals.
उत्तर २
Actinoids are far more reactive than lanthanoids. They interact with nonmetals at moderate temperatures. In contrast, lanthanoids react at high temperatures. The majority of actinoids are attacked by HNO3 has a small effect on HCl, although the creation of a protective layer of oxide. Alkalies cause no reaction. Lanthanoids extract hydrogen from dilute. Acids and halogens burn together to generate halides.
APPEARS IN
संबंधित प्रश्न
Name a member of the lanthanoid series which is well known to exhibit +4 oxidation state.
Name the members of the lanthanoid series which exhibit +4 oxidation state and those which exhibit +2 oxidation state. Try to correlate this type of behavior with the electronic configurations of these elements.
Write the electronic configuration of the element with the atomic number 61.
Write the different oxidation states of iron
Explain, why lanthanum (Z = 57) forms La3+ ion, while cerium (Z = 58) forms Ce4+ ion?
Explain effects of lanthanoid contraction.
Account for the following:
Zn, Cd and Hg are soft metals.
Which trivalent ion has the maximum size in the Lanthanoid series, i.e., Lanthanum ion (La3+) to Luteium ion (Lu3+)?
(at. no. of Lanthanum = 57 and Lutetium = 71)
Gadolinium belongs to 4f series. It’s atomic number is 64. Which of the following is the correct electronic configuration of gadolinium?
General electronic configuration of actinoids is `(n-2)f^(1-14)(n - 1)d^(0-2)ns^2`.Which of the following actinoids have one electron in 6d orbital?
(i) U (Atomic no. 92)
(ii) Np (Atomic no.93)
(iii) Pu (Atomic no. 94)
(iv) Am (Atomic no. 95)
Although +3 oxidation states is the characteristic oxidation state of lanthanoids but cerium shows +4 oxidation state also. Why?
Match the property given in Column I with the element given in Column II.
| Column I (Property) | Column II (Element) | |
| (i) | Lanthanoid which shows +4 oxidation state |
(a) Pm |
| (ii) | Lanthanoid which can show +2 oxidation state |
(b) Ce |
| (iii) | Radioactive lanthanoid | (c) Lu |
| (iv) | Lanthanoid which has 4f7 electronic configuration in +3 oxidation state |
(d) Eu |
| (v) | Lanthanoid which has 4f14 electronic configuration in +3 oxidation state |
(e) Gd |
| (f) Dy |
On the basis of Lanthanoid contraction, explain the following:
Stability of the complexes of lanthanoids.
On the basis of Lanthanoid contraction, explain the following:
Trends in acidic character of lanthanoid oxides.
State a reason for the following:
La(OH)3 is more basic than Lu(OH)3.
Assertion: There is a continuous increase in size among Lanthanoids with an increase in atomic number.
Reason: Lanthanoids do not show Lanthanoid contraction.
Name an important alloy which contains some of the lanthanoid metals. Mention its uses.
The pair of lanthanoid ions which are diamagnetic is:
