Advertisements
Advertisements
प्रश्न
Classify the following compound as a primary, secondary and tertiary halide.
1-Bromobut-2-ene
विकल्प
Primary halide
Secondary halide
Tertiary halide
Advertisements
उत्तर
Primary halide
Explanation:
\[\ce{\underset{1-Bromobut-2-ene}{CH3HC = CHCH2Br}}\]
APPEARS IN
संबंधित प्रश्न
Draw the structure of the major monohalo product in the following reaction:

Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)2CHCH(Cl)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2C(CH3)2CH2I
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH = C(Cl)CH2CH(CH3)2
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
p-ClC6H4CH2CH(CH3)2
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(a) Aryl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (b) Alkyl halide |
| (iii) | 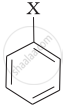 |
(c) Vinyl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (d) Allyl halide |
Which alkyl halide has maximum density
Which of the following belongs to the class of vinyl halides?
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2 C(CH3)2CH2I}\]
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2C(CH3)2CH2I
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3CH2C(CH3)2CH2l
Give the IUPAC name of the compound.
\[\ce{CICH2 C ≡ CCH2 Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
IUPAC name of \[\begin{array}{cc}
\phantom{}\ce{CH3}\phantom{..........}\ce{Br}\phantom{........}\\
\phantom{}|\phantom{................}|\phantom{.........}\\
\phantom{}\ce{CH3CHCH2CHCHCH2CH3}\phantom{}\\
\phantom{}|\phantom{..}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\] is ______.
IUPAC name of 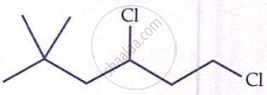 is ______.
is ______.
Which is the correct IUPAC name for 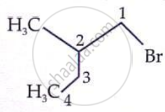 is______.
is______.
