Advertisements
Advertisements
प्रश्न
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
विकल्प
Potassium nitrate
Slacked lime
Quick lime
Sodium nitrate
Advertisements
उत्तर
Slacked lime
APPEARS IN
संबंधित प्रश्न
Is ammonia more dense or less dense than air?
When an ammonium salt is warmed with a sodium hydroxide solution, a gas is evolved. State three ways in which you can identify this gas.
Give the formula of (i) Liquid ammonia (ii) Liquor ammonia
Mention two possible ways by which ammonia produced is removed from unchanged gases.
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
Given one test can be used to detect the presence of the ion produced
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
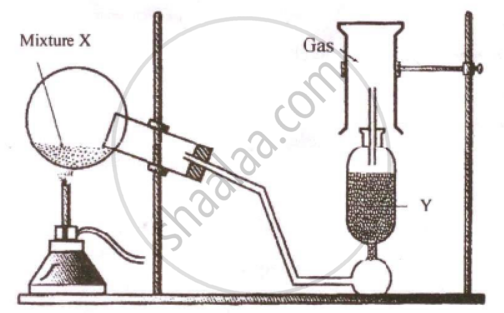
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Ammonium solution is added to ferric chloride solution.
How is ammonia soluble in water?
Identify the terms for the following:
The experiment which demonstrates high solubility of ammonia gas.
