Advertisements
Advertisements
प्रश्न
Chloromethane on treatment with excess of ammonia yields mainly ______.
विकल्प
N, N-Dimethylmethanamine
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\phantom{}\\
\phantom{.........}/{}\phantom{}\\
\phantom{}\ce{CH3 - N\phantom{..}\\
\phantom{.........}\backslash{}\phantom{}\\
\phantom{..............}\ce{CH3}\phantom{}\\
\ce{}}
\end{array}\]N–methylmethanamine (CH3—NH—CH3)
Methenamine (CH3NH2)
Mixture containing all these in equal proportion
Advertisements
उत्तर
Chloromethane on treatment with excess of ammonia yields mainly Methenamine (CH3NH2).
Explanation:
Chloromethane on treatment with excess of ammonia yields mainly methenamine.
\[\ce{CH3Cl + NH3 –> CH3NH2 + HCl}\]
Excess Methanamine
\[\ce{CH3Cl + \underset{Excess}{NH3} -> \underset{methenamine}{CH3NH2} + HCl}\]
However, if the two reactants are present in the same amount, a mixture of the primary, secondary and tertiary amine is obtained.
\[\ce{CH3Cl + NH3 -> \underset{(Primary amine)}{CH3NH2} + HCl}\]
\[\ce{CH3NH2 + CH3Cl -> \underset{(Secondary amine)}{(CH3)2NH}+ HCl}\]
\[\ce{(CH3)2NH + CH3Cl -> \underset{(Tertiary amine)}{(CH3)3N} + HCl}\]
\[\ce{(CH3)2NH + CH3Cl -> \underset{(Quarternary ammonium salt)}{(CH3)4 \overset{+}{N} \overset{-}{Cl}}}\]
APPEARS IN
संबंधित प्रश्न
Give the uses of freon 12.
How can the following conversions be brought about:
Methyl chloride to acetone.
Give balanced equations for the following reaction:
Acetaldehyde with hydroxylamine.
Write the formula of the following compound:
Triammine triaquachromium (III) chloride
Give a balanced chemical equation to convert methyl cyanide to ethyl alcohol.
In the following sequence of the reactions, what is D?
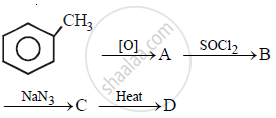
In which of the following conversions, phosphorus pentachloride is used as the reagent?
Chloroform and conc. HNO3 reacts to produce ____________.
What are the IUPAC names of the insecticide DDT and benzene hexachloride? Why is their use banned in India and other countries?
Convert the following:
Chloroethane to methanamine.
The most stable conformation of 1, 2-dibromomethane among the following is
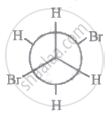 |
| I |
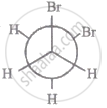 |
| II |
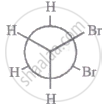 |
| III |
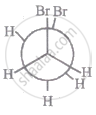 |
| IV |
Ethyl bromide reacts with lead sodium alloy to from:-
Chloroform on reduction with Zn an HCI (all) gives:-
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
Write equation to convert Methyl cyanide to ethanoic acid.
Strong reducing agent converts CHCl3 into:
Assertion (A): Exposure of ultraviolet rays to human causes the skin cancer, disorder and disrupt the immune system. Sol. Reason (R): Carbon tetrachloride is released into air it rises to atmosphere and deplets the ozone layer. In the light of above statements, choose the most appropriate answer from the options given below.
Iodoform can be prepared from all except:
