Advertisements
Advertisements
प्रश्न
Write the environmental effects of DDT.
Advertisements
उत्तर
- It is not completely biodegradable i.e. it is not very rapidly metabolized by animals. Instead, it gets deposited and store in fatty tissues. It is exists for longtime in soil, plants and animals as well. It produces unforeseen ecological effects.
- It is replaced by better and safer insecticide due to ecological problems.
APPEARS IN
संबंधित प्रश्न
Draw structure of DDT.
Give the uses of freon 12.
Give balanced equations for the following reaction:
Acetaldehyde with hydroxylamine.
Write balanced chemical equation for the following reaction:
Methyl magnesium iodide is treated with carbon dioxide and the product hydrolysed in acidic medium.
Write the formula of the following compound:
Triammine triaquachromium (III) chloride
Give a balanced chemical equation to convert methyl cyanide to ethyl alcohol.
In the following sequence of the reactions, what is D?
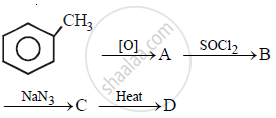
In which of the following conversions, phosphorus pentachloride is used as the reagent?
Chloroform and conc. HNO3 reacts to produce ____________.
B.H.C. is used as an ____________.
Gammexane is ____________.
Chloropicrin is formed by the reaction of ____________.
Fitting reaction can be used to prepare ____________.
A dihalogen derivative ‘X’ of a hydrocarbon with three carbon atoms react with ale. KOH and produces hydrocarbon which forms red ppt. with ammonical Cu2Cl2. ‘X’ gives an aldehyde on reaction with aq. KOH. The compound ‘X’ is:
Major product obtained on reaction of 3-Phenyl propene with HBr in presence of organic peroxide.
Why iodoform has appreciable antiseptic property?
Name the alkene which will yield 1-chloro-1-methylcyclohexane by its reaction with \[\ce{HCl}\]. Write the reactions involved.
How will you obtain monobromobenzene from aniline?
How can you obtain iodoethane from ethanol when no other iodine-containing reagent except NaI is available in the laboratory?
Match the compounds given in Column I with the effects given in Column II.
| Column I | Column II | |
| (i) | Chloramphenicol | (a) Malaria |
| (ii) | Thyroxine | (b) Anaesthetic |
| (iii) | Chloroquine | (c) Typhoid fever |
| (iv) | Chloroform | (d) Goiter |
| (e) Blood substituent |
Assertion: It is difficult to replace chlorine by –OH in chlorobenzene in comparison to that in chloroethane.
Reason: Chlorine-carbon \[\ce{(C - Cl)}\] bond in chlorobenzene has a partial double bond character due to resonance.
The most stable conformation of 1, 2-dibromomethane among the following is
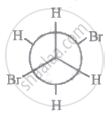 |
| I |
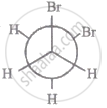 |
| II |
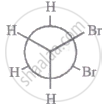 |
| III |
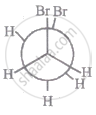 |
| IV |
Which of the following does not give iodoform test?
The shape of methyl carbanion is similar to that of–
Ethyl chloride is converted into diethyl ether by
Chlorination of CS2 give
Sodium ethoxide react with ethyl iodide to yield.
CH3NH2 react with CH3mg x to give
Chloroform on reduction with Zn an HCI (all) gives:-
Westrosol is:-
Which among the following pairs has only herbicides?
The major product formed when 1, 1, 1-trichloropropane is treated with aqueous potassium hydroxide, is ______.
State the uses of iodoform (CHI3)
Write names of two polyhalogen compounds.
Convert the following iodomethane to methoxy methane.
Write equation to convert Methyl cyanide to ethanoic acid.
Give the uses of DDT.
Give the uses of carbon tetrachloride.
Give the uses of iodoform.
DDT is obtained by the reaction of chlorobenzene with ______.
Methyl bromide is not used ______.
Strong reducing agent converts CHCl3 into:
Assertion (A): Exposure of ultraviolet rays to human causes the skin cancer, disorder and disrupt the immune system. Sol. Reason (R): Carbon tetrachloride is released into air it rises to atmosphere and deplets the ozone layer. In the light of above statements, choose the most appropriate answer from the options given below.
Assertion (A): CHCI3 is stored in dark bottles. Reason (R): CHCl3 is oxidised in dark.
In the light of above statements, choose the most appropriate answer from the options given below
Which of these compounds is synthesised by chloral?
