Advertisements
Advertisements
प्रश्न
Answer the following in brief.
How will you determine activation energy graphically using the Arrhenius equation?
Advertisements
उत्तर
Graphical representation of activation energy
Arrhenius equation is `"k" = "Ae"^((-"E"_"a")/("RT"))`
Taking the logarithm of both sides of Eqn. we obtain
In k = -`"E"_"a"/"RT" + "In A"`
Converting the natural base to base 10 we write
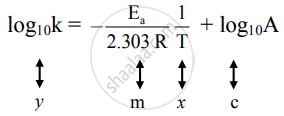
This equation is of the form of straight-line y = mx + c.
The Arrhenius plot of log10k versus `1/"T"` gives a straight line as shown in the diagram. A slope of the line is `-"E"_"a"/(2.303 "R")` with its intercept being log10A.
From a slope of the line, the activation energy can be determined.
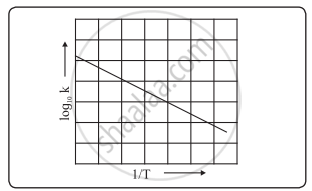
Variation of log10k with 1/T
APPEARS IN
संबंधित प्रश्न
Explain with the help of the Arrhenius equation, how do the rate of reaction changes with temperature.
Answer the following in brief.
Explain graphically the effect of temperature on the rate of reaction.
Solve
The energy of activation for a first-order reaction is 104 kJ/mol. The rate constant at 25°C is 3.7 × 10–5 s –1. What is the rate constant at 30°C? (R = 8.314 J/K mol)
Solve
What is the energy of activation of a reaction whose rate constant doubles when the temperature changes from 303 K to 313 K?
How will you determine activation energy from rate constants at two different temperatures?
Explain with the help of the Arrhenius equation, how do the rate of reaction changes with activation energy.
A catalyst lowers the activation energy of a certain reaction from 83.314 to 75 kJ mol−1 at 500 K. What will be the rate of reaction as compared to uncatalysed reaction? Assume other things are equal.
Slope of the straight line obtained by plotting log10k against represents what term?
Explain with the help of Arrhenius equation, how does the rate of reaction changes with activation energy.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with activation energy.
Explain, with the help of the Arrhenius equation, how does the rate of reaction changes with temperature.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with temperature.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with temperature.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with activation energy.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with temperature.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with temperature.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with activation energy.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with temperature.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with activation energy.
Explain with the help of Arrhenius equation, how does the rate of reaction changes with temperature.
The formation of micelles takes place only above ______.
