Advertisements
Advertisements
प्रश्न
Account for the following :
t-butyl chloride on heating with sodium methoxide gives 2-methylpropene instead of t-butyl methyl ether.
Advertisements
उत्तर
Sodium methoxide is a strong base hence elimination predominates over substitution
\[\begin{array}{cc}
\ce{CH3}\phantom{.............................}\\
|\phantom{...............................}\\
\ce{CH3-C-Cl +CH3O^-Na^+->CH3-C=CH3OH + NaCI}\\
|\phantom{...............................}||\\
\phantom{....}\ce{CH3}\phantom{..............}\phantom{..............}\ce{CH2}
\end{array}\]
William synthesis
\[\ce{R - X + R -\underset{\bullet\bullet}{{}^{\bullet\bullet}_{O}} - Na->R - \underset{\bullet\bullet}{{}^{\bullet\bullet}_{O}} - R + NaX}\]
The alkyl halide must be 1⁰ as the reaction involves the SN2 reaction pathway.
If alkyl halide is 3⁰ then the alkene will be the major product. Hence, 2-methylpropene is formed instead of t-butylmethylether.
संबंधित प्रश्न
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
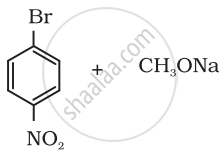 |
 |
| (i) | (ii) |
\[\ce{(CH3)3CONa + CH3CH2Cl ->[-NaCl] (CH3)3COC2H5}\] is called:
Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism.
Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
The major product [B] in the following reactions is:
\[\begin{array}{cc}\ce{CH3}\phantom{..................................}\\|\phantom{.....................................}\\\ce{CH3 - CH2 - CH - CH2 - OCH2 - CH3 ->[HI][Heat] [A] alcohol ->[H2SO4][\Delta] [B]}\end{array}\]
Williamson's synthesis of ether is an example of ______.
Write the mechanism of the following reaction:
\[\ce{2CH3CH2OH ->[H^+][413 K] CH3-CH2-O-CH2-CH3 + H2O}\]
Give the structure and IUPAC name of metamers of 2-methoxy propane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
