Advertisements
Advertisements
प्रश्न
A solution contains 5.6 mL of alcohol mixed with 75 mL of water. Calculate the concentration of this solution.
Advertisements
उत्तर
Volume of alcohol = 5.6 ml
Volume of water = 75 ml
So, volume of solution = 75ml + 5.6 ml = 80.6 ml
`"concentration"= "volume of solute"/"volume of solute + volume of solvent"xx100`
`= 5.6/80.6xx100`
= 6.9%
APPEARS IN
संबंधित प्रश्न
The molecules can move freely anywhere in
Fill in the blank :
The state of matter with definite volume and definite shape is called______
Name the terms for the change of a solid into liquid.
Honey is more viscous than water. Can you suggest why?
Explain why air is used to inflate tyres.
Which of the following will show Tyndall effect ? Why ?
- Salt solution
- Starch solution
- Milk
- Copper sulphate solution
What do you understand by the statement " the solubility of copper sulphate in water at 20°C is 20.7 g" ?
Milk of Magnesia is :
How is the impurity of iron present in several substances removed in industries ?
A solid mixture contains four constituents P, Q, R and S. P consists of tiny grains and it is mixed with
cement for plastering the walls. Q is a white solid which is recovered on a large scale from sea water by the
process of evaporation. R is in the form of tiny particles of a material whose corrosion is called rusting. And
S is a white solid which is used in making ordinary dry cells.
- What could P, Q, R and S be ?
- How would you separate a mixture containing P, Q, R and S ?
What's the solution?
We need to make a sherbet quickly for some guests. But we only have sugar candy in the house.
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
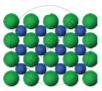 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Why wires cannot be drawn from materials such as stone or wood?
Stars including Sun are covered in ______.
Seema visited a Natural Gas Compressing Unit and found that the gas can be liquefied under specific conditions of temperature and pressure. While sharing her experience with friends she got confused. Help her to identify the correct set of conditions
Complete the following table:
| S.No. | CELSIUS | KELVIN |
| 1. | 90°C | 363 K |
| 2. | ______ | 283 K |
| 3. | 63 °C | ______ |
| 4. | 250°C | ______ |
| 5. | ______ | 303 K |
The physical state of water at 373 K is ______.
Boiling refers to the process by which a substance changes from the liquid to the gaseous state at its ______ point.
Define the term matter.
Define the term matter.
