Advertisements
Advertisements
प्रश्न
A 12.5 eV electron beam is used to bombard gaseous hydrogen at room temperature. Upto which energy level the hydrogen atoms would be excited? Calculate the wavelengths of the first member of Lyman and first member of Balmer series.
Advertisements
उत्तर
Energy of the electron in the nth state of an atom `=-(13.6z^2)/n^2eV`
Here, z is the atomic number of the atom.
For hydrogen atom, z is equal to 1.
Energy required to excite an atom from the initial state (ni) to the final state (nf) = `-13.65/(nf^2)+13.6/n_(i^2)eV`
This energy must be equal to or less than the energy of the incident electron beam.
`:.-13.6/(nf^2)+13.6/n_(i^2)=12.5`
Energy of the electron in the ground state = `-13.6/1^2=-13.6V`
`:.-13.6/nf^2+13.6=12.5`
`=>13.6-12.5=13.6/(nf^2)`
`=>nf^2=13.6/1.1=12.36`
`=>n_f=3.5`
State cannot be a fraction number.∴ nf = 3
Hence, hydrogen atom would be excited up to 3rd energy level.
Rydberg formula for the spectrum of the hydrogen atom is given below:
` 1/lambda=R[1/n_(1^2)-1/n_(2^2)]`
Here, λ is the wavelength and R is the Rydberg constant.
R = 1.097 × 107 m-1
For the first member of the Lyman series:
n1 = 1
n2 = 2
`1/lambda=1.097xx10^7[1/1^2-1/2^2]`
λ = 1215 Å
For the first member of Balmer series:
n1 = 2
n2 = 3
`1/lambda=1.097xx10^7[1/2^2-1/3^2]`
⇒λ = 6563 Å
APPEARS IN
संबंधित प्रश्न
Given the ground state energy E0 = - 13.6 eV and Bohr radius a0 = 0.53 Å. Find out how the de Broglie wavelength associated with the electron orbiting in the ground state would change when it jumps into the first excited state.
The total energy of an electron in the first excited state of the hydrogen atom is about −3.4 eV.
What is the kinetic energy of the electron in this state?
What are means by pair annihilation? Write a balanced equation for the same.
A 12.3 eV electron beam is used to bombard gaseous hydrogen at room temperature. Upto which energy level the hydrogen atoms would be excited?
Calculate the wavelengths of the second member of Lyman series and second member of Balmer series.
Draw the energy level diagram showing how the line spectra corresponding to Paschen series occur due to transition between energy levels.
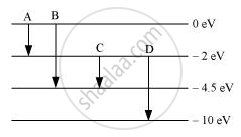
The energy levels of an atom are as shown below. Which of them will result in the transition of a photon of wavelength 275 nm?
A Carnot engine absorbs 1000 J of heat energy from a reservoir at 127°C and rejects 600 J of heat energy during each cycle. The efficiency of the engine and temperature of the sink will be:
Two H atoms in the ground state collide inelastically. The maximum amount by which their combined kinetic energy is reduced is ______.
Which of the following is true for X-rays?
The diagram shows the four energy levels of an electron in the Bohr model of the hydrogen atom. Identify the transition in which the emitted photon will have the highest energy.

