Advertisements
Advertisements
Can we use nucleophiles such as NH3, CH3O for the Nucleophilic substitution of alcohols?
Concept: undefined >> undefined
Is it possible to oxidise t-butyl alcohol using acidified dichromate to form a carbonyl compound?
Concept: undefined >> undefined
Advertisements
What happens when 1-phenyl ethanol is treated with acidified KMnO4.
Concept: undefined >> undefined
Write the mechanism of acid catalysed dehydration of ethanol to give ethene.
Concept: undefined >> undefined
How is phenol prepared from isopropyl benzene?
Concept: undefined >> undefined
Explain Kolbe’s reaction.
Concept: undefined >> undefined
Write the chemical equation for Williamson's synthesis of 2-ethoxy-2-methyl pentane starting from ethanol and 2-methyl pentan-2-ol.
Concept: undefined >> undefined
Write the structure of the aldehyde, carboxylic acid and ester that yield 4-methylpent-2-en-1-ol.
Concept: undefined >> undefined
How is the conversion effected benzyl chloride to benzyl alcohol?
Concept: undefined >> undefined
How is the conversion effected benzyl alcohol to benzoic acid?
Concept: undefined >> undefined
Complete the following reaction.
\[\ce{C6H5 - OH ->[Zn dust] A ->[CH3Cl][Anhydrous AlCl3] B ->[acid KMnO4] C}\]
Concept: undefined >> undefined
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
Concept: undefined >> undefined
Complete the following reaction.
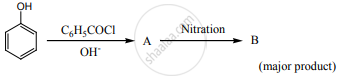
Concept: undefined >> undefined
Complete the following reaction.
\[\ce{C6H5 - CH2CH(OH)CH(CH3)2 ->[conc. H2SO4]}\]
Concept: undefined >> undefined
Phenol is distilled with Zn dust followed by Friedel-crafts alkylation with propyl chloride to give a compound B, B on oxidation gives (c) Identify A, B and C.
Concept: undefined >> undefined
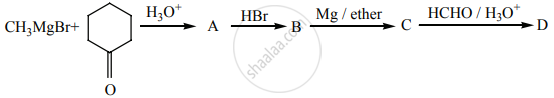
Identify A, B, C, D and write the complete equation.
Concept: undefined >> undefined
What will be the product (X and A) for the following reaction?
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
Concept: undefined >> undefined
How will you convert acetylene into n-butyl alcohol?
Concept: undefined >> undefined
3, 3-dimethyl butane-2-ol on treatment with conc. H2SO4 to give tetramethyl ethylene as a major product. Suggest a suitable mechanism.
Concept: undefined >> undefined
The IUPAC name of

Concept: undefined >> undefined
