Advertisements
Advertisements
Give a plausible explanation for the following:
Why are aliphatic amines stronger bases than aromatic amines?
Concept: undefined >> undefined
Complete the following reaction:
\[\ce{C6H5N2Cl + H3PO2 + H2O ->}\]
Concept: undefined >> undefined
Advertisements
Write the structure of the product of the following reaction:

Concept: undefined >> undefined
Predict the product of the following reaction:
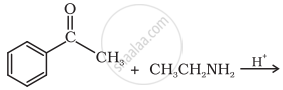
Concept: undefined >> undefined
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzaldehyde
Concept: undefined >> undefined
Give the equations of reactions for the preparation of phenol from cumene.
Concept: undefined >> undefined
Write chemical reaction for the preparation of phenol from chlorobenzene.
Concept: undefined >> undefined
You are given benzene, conc. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
Concept: undefined >> undefined
Write the equation for the preparation of 1-iodobutane from but-1-ene.
Concept: undefined >> undefined
How will you bring about the following conversion?
Ethane to bromoethene
Concept: undefined >> undefined
How the following conversion can be carried out?
Aniline to chlorobenzene
Concept: undefined >> undefined
What happens when white phosphorus is heated with concentrated NaOH solution in an inert atmosphere of CO2?
Concept: undefined >> undefined
Explain effects of lanthanoid contraction.
Concept: undefined >> undefined
What are the consequences of lanthanoid contraction?
Concept: undefined >> undefined
Write the IUPAC name of the following coordination compound:
K3[Fe(CN)6]
Concept: undefined >> undefined
Write the IUPAC names of the following coordination compounds:
[CoBr2(en)2]+, (en = ethylenediamine)
Concept: undefined >> undefined
Identify that the following compound is insoluble, partially soluble and highly soluble in water.
phenol
Concept: undefined >> undefined
Identify that the following compound is insoluble, partially soluble and highly soluble in water.
Toluene
Concept: undefined >> undefined
Identify that the following compound is insoluble, partially soluble or highly soluble in water:
Formic acid
Concept: undefined >> undefined
Identify that the following compound is insoluble, partially soluble or highly soluble in water:
Ethylene glycol
Concept: undefined >> undefined
