Please select a subject first
Advertisements
Advertisements
Elements are broadly classified into metals and non-metals. State six general differences in the physical properties of metals and non-metals. State two metals and two non-metals which contradict with the general physical properties – giving reasons. State one difference in property between metalloids and noble gases.
Concept: undefined >> undefined
With reference to elements – define the term ‘molecule’. Give two examples each of a monoatomic, diatomic & polyatomic molecule.
Concept: undefined >> undefined
Advertisements
Define the term ‘compound’. In the compound carbon dioxide – the elements carbon & oxygen are combined in a fixed ratio. Explain.
Concept: undefined >> undefined
State five different characteristics of compounds.
Concept: undefined >> undefined
Give three differences between elements & compounds with relevant examples.
Concept: undefined >> undefined
Explain the term ‘mixture’.
Concept: undefined >> undefined
Differentiate between homogenous & heterogeneous mixtures.
Concept: undefined >> undefined
State why brass is considered as a homogenous mixture while a mixture of iron & sulphur – heterogenous.
Concept: undefined >> undefined
Give an example of two liquids which form homogenous.
Concept: undefined >> undefined
Give an example of two liquids that form heterogeneous mixtures.
Concept: undefined >> undefined
Select the correct answer from A, B, C, D & E for the statement given below :
A diatomic molecule.
Concept: undefined >> undefined
Select the correct answer from A, B, C, D & E for the statement given below :
A metalloid.
Concept: undefined >> undefined
Select the correct answer from A, B, C, D & E for the statement given below :
A non-metal that is lustrous.
Concept: undefined >> undefined
Select the correct answer from A, B, C, D & E for the statement given below :
A mixture consisting of elements & a compound.
Concept: undefined >> undefined
Select the correct answer from A, B, C, D & E for the statement given below :
A noble gas.
Concept: undefined >> undefined
Match the separation of components in List I with the most appropriate process in List II.
| List I | List II |
| 1. Naphthalene from naphthalene & sodium chloride. | A: Separating funnel |
| 2. Cream from milk. | B: Sublimation |
| 3. Kerosene oil from kerosene oil & water. | C: Boiling |
| 4. Lead nitrate from an aqueous solution of lead nitrate. | D: Centrifugation |
| 5. Ammonia from an aqueous solution of ammonia. | E: Distillation |
Concept: undefined >> undefined
The diagram represents fractional distillation for the separation of mixtures. Answer the following :
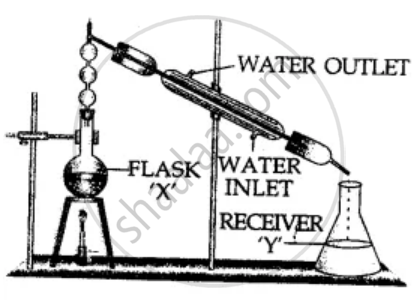
- Can two immiscible liquids be separated by this process?
- Separation of liquids by this process is based on which physical property?
- If methyl alcohol & water are to be separated, which liquid would remain in flask ‘X’ after condensation.
- Give a reason for the above answer.
- State the purpose of the fractionating column in the apparatus.
Concept: undefined >> undefined
Select the correct answer from the choice given below to complete the sentence :
Dust in air is an example of _________ mixture.
Concept: undefined >> undefined
Select the correct answer from the choice given below to complete the sentence :
Compounds are ____________ in nature.
Concept: undefined >> undefined
Select the correct answer from the choice given below to complete the sentence :
An example of a monoatomic molecule is ___________.
Concept: undefined >> undefined
