Advertisements
Advertisements
Describe two tests to identify ammonia and ammonium ions in an aqueous solution.
Concept: undefined >> undefined
Concept: undefined >> undefined
Advertisements
Concept: undefined >> undefined
Concept: undefined >> undefined
Choose the correct word or phrase from the bracket to complete the following sentence:
Heating ammonium chloride with sodium hydroxide produces ______.
Concept: undefined >> undefined
Concept: undefined >> undefined
Concept: undefined >> undefined
Choose the correct word or phrase from the bracket to complete the following sentences.
Heating solution of ammonium chloride with sodium nitrite produces ______
Concept: undefined >> undefined
Concept: undefined >> undefined
Concept: undefined >> undefined
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
Concept: undefined >> undefined
The diagram shows a simple arrangement of the fountain experiment:

(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
Concept: undefined >> undefined
Name the other ion formed when ammonia dissolves in water
Concept: undefined >> undefined
Name the gas evolved when the following mixtures are heated.
Calcium hydroxide and ammonium chloride
Concept: undefined >> undefined
Name the gas evolved when the following mixtures are heated:
Sodium nitrate and ammonium chloride.
Concept: undefined >> undefined
Identify the substance underlined, in the following case:
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide.
Concept: undefined >> undefined
Identify the substance underlined, in the following case:
A solid formed by reaction of two gaes, one of which is acidic and the other basic in nature.
Concept: undefined >> undefined
State one relevant observation of burning of ammonia in air.
Concept: undefined >> undefined
Given one test can be used to detect the presence of the ion produced
Concept: undefined >> undefined
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
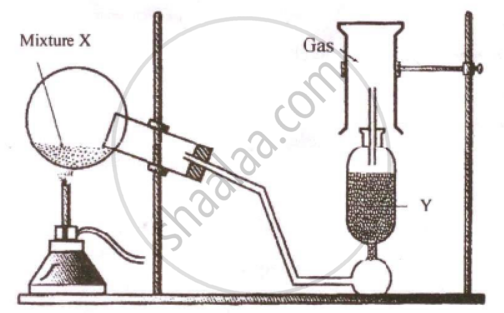
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
Concept: undefined >> undefined
