Advertisements
Advertisements
प्रश्न
Name the gas evolved when the following mixtures are heated.
Calcium hydroxide and ammonium chloride
एक शब्द/वाक्यांश उत्तर
Advertisements
उत्तर
Ammonia
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
संबंधित प्रश्न
Ammonium salts decompose on heating. What other property do ammonium salts have in common?
Give two reactions to show reducing property of ammonia.
Name a metallic chloride soluble in ammonium hydroxide.
Complete the following equation. What property of ammonia is illustrated by the reaction given below.
2NH3 + 3CuO →
What is liquor ammonia fortis?
How will you demonstrate the solubility of ammonia in water? Explain.
Distinguish between liquid ammonia and liquid ammonia fortis.
Name the gas evolved when the following mixtures are heated:
Sodium nitrate and ammonium chloride.
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
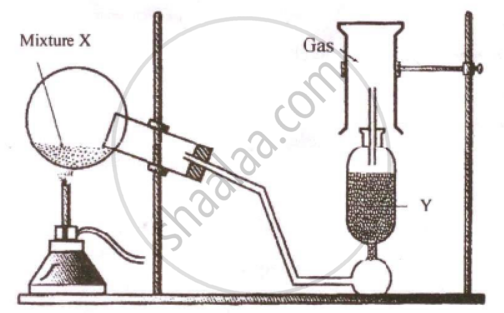
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
- Of the two gases, ammonia and hydrogen chloride, which is more dense? Name the method of collection of this gas.
- Give one example of a reaction between the above two gases which produce a solid compound.
- Write the balanced equation for a reaction in which ammonia is oxidized by:
- a metal oxide,
- a gas which is not oxygen.
