Advertisements
Advertisements
1-Propanol and 2-propanol can be best distinguished by:
Concept: undefined >> undefined
Which of the following is most acidic?
Concept: undefined >> undefined
Advertisements
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
Concept: undefined >> undefined
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
Concept: undefined >> undefined
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
Concept: undefined >> undefined
The product of acid catalysed hydration of 2-phenylpropene is:
Concept: undefined >> undefined
NaCl type crystal (with coordination no. 6 : 6) can be converted into CsCl type crystal (with coordination no. 8 : 8) by applying ____________.
Concept: undefined >> undefined
How many chloride ions are surrounding sodium ions in sodium chloride crystal?
Concept: undefined >> undefined
The number of moles of NaCl in 3 litres of 3 M solution is ____________.
Concept: undefined >> undefined
4 L of 0.02 M aqueous solution of NaCl was diluted by adding one litre of water. The molality of the resultant solution is ______.
Concept: undefined >> undefined
Cone. H2SO4 is 98% H2SO4 by mass has d = 1.84 g cm−3. Volume of acid required to make one litre of 0.1 M H2SO4 is:
Concept: undefined >> undefined
What is the mole fraction of solute in 1.00 m aqueous solutions?
Concept: undefined >> undefined
\[\begin{array}{cc}
\ce{CH3-CH-CH=CH2 + HBr -> A}\\
|\phantom{.....................}\\
\ce{CH3}\phantom{..................}
\end{array}\]
'A' is:
Concept: undefined >> undefined
The heating of phenyl methyl ether with HI produces:
Concept: undefined >> undefined
Which of the following gives a positive iodoform test?
Concept: undefined >> undefined
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Concept: undefined >> undefined
Among the following sets of reactants which one produces anisole?
Concept: undefined >> undefined
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
Concept: undefined >> undefined
The correct acidic strength order of the following is:

(I)
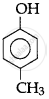
(II)

(III)
Concept: undefined >> undefined
Which of the following is a correct statement for C2H5Br?
Concept: undefined >> undefined
