Please select a subject first
Advertisements
Advertisements
Which technique utilizes the density difference between the ore and the impurity to concentrate the ore is:
Concept: undefined >> undefined
The diagram shows a schematic representation of
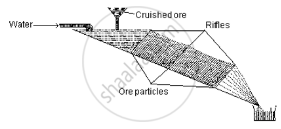
Concept: undefined >> undefined
Advertisements
In laboratory ammonia is prepared by heating:
Concept: undefined >> undefined
Ammonia has a higher boiling point and is less volatile because of ____________.
Concept: undefined >> undefined
Which compound is used in the preparation of caprolactam?
Concept: undefined >> undefined
Which compound is used as the cooling liquid in refrigerators?
Concept: undefined >> undefined
Ammonia act as a Lewis base because nitrogen has ____________.
Concept: undefined >> undefined
Which among the following elements does not belong to the first transition series?
Concept: undefined >> undefined
Which among the following is not considered as a part of transition elements?
Concept: undefined >> undefined
Electronic configuration of Cu3+ is ____________.
Concept: undefined >> undefined
Which of the following represents a chelate ligand?
Concept: undefined >> undefined
Ligand (en) is an example of ___________.
Concept: undefined >> undefined
A group of atoms can function as a ligand only when:
Concept: undefined >> undefined
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
Concept: undefined >> undefined
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
Concept: undefined >> undefined
The coordination number of the central ion may be obtained from:
Concept: undefined >> undefined
In which of the following compounds the oxidation state of the nickel atom is 0?
Concept: undefined >> undefined
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Concept: undefined >> undefined
Which of the following is non-ionizable?
Concept: undefined >> undefined
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Concept: undefined >> undefined
