Advertisements
Advertisements
Rationalise the given statement and give a chemical reaction:
Lead (IV) chloride is highly unstable towards heat.
Concept: undefined >> undefined
Rationalise the given statement and give a chemical reaction:
Lead is known not to form an iodide, PbI4.
Concept: undefined >> undefined
Advertisements
Classify the following oxide as neutral, acidic, basic or amphoteric:
CO
Concept: undefined >> undefined
Classify the following oxide as neutral, acidic, basic or amphoteric:
B2O3
Concept: undefined >> undefined
Classify the following oxide as neutral, acidic, basic or amphoteric:
SiO2
Concept: undefined >> undefined
Classify the following oxide as neutral, acidic, basic or amphoteric:
CO2
Concept: undefined >> undefined
Classify the following oxide as neutral, acidic, basic or amphoteric:
Al2O3
Concept: undefined >> undefined
Classify the following oxide as neutral, acidic, basic or amphoteric:
PbO2
Concept: undefined >> undefined
Classify the following oxide as neutral, acidic, basic or amphoteric:
Tl2O3
Concept: undefined >> undefined
Concept: undefined >> undefined
Write suitable chemical equations to show the nature of the following oxide.
B2O3
Concept: undefined >> undefined
Write suitable chemical equations to show the nature of the following oxide.
SiO2
Concept: undefined >> undefined
Write suitable chemical equations to show the nature of the following oxide.
CO2
Concept: undefined >> undefined
Write suitable chemical equations to show the nature of the following oxide.
Al2O3
Concept: undefined >> undefined
Write suitable chemical equations to show the nature of the following oxide.
PbO2
Concept: undefined >> undefined
Name the energy which arises due to motion of atoms or molecules in a body. How is this energy affected when the temperature is increased?
Concept: undefined >> undefined
The pressure-volume work for an ideal gas can be calculated by using the expression w = `- int_(v_i)^(v_f) p_(ex) dV`. The work can also be calculated from the pV– plot by using the area under the curve within the specified limits. When an ideal gas is compressed (a) reversibly or (b) irreversibly from volume Vi to Vf. choose the correct option.
Concept: undefined >> undefined
For an ideal gas, the work of reversible expansion under isothermal condition can be calculated by using the expression w = `- nRT` In `V_f/V_i`. A sample containing 1.0 mol of an ideal gas is expanded isothermally and reversibly to ten times of its original volume, in two separate experiments. The expansion is carried out at 300 K and at 600 K respectively.
(i) Work done at 600 K is 20 times the work done at 300 K.
(ii) Work done at 300 K is twice the work done at 600 K.
(iii) Work done at 600 K is twice the work done at 300 K.
(iv) ∆U = 0 in both cases.
Concept: undefined >> undefined
A sample of 1.0 mol of a monoatomic ideal gas is taken through a cyclic process of expansion and compression as shown in figure 6.1. What will be the value of ∆H for the cycle as a whole?
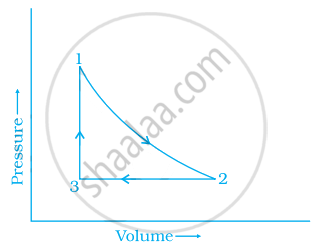
Concept: undefined >> undefined
Expansion of a gas in vacuum is called free expansion. Calculate the work done and the change in internal energy when 1 litre of ideal gas expands isothermally into vacuum until its total volume is 5 litre?
Concept: undefined >> undefined
