Please select a subject first
Advertisements
Advertisements
The entropy change can be calculated by using the expression ∆S = `q_(rev)/T`. When water freezes in a glass beaker, choose the correct statement amongst the following :
Concept: undefined >> undefined
The spontaneity means, having the potential to proceed without the assistance of external agency. The processes which occur spontaneously are:
(i) Flow of heat from colder to warmer body.
(ii) Gas in a container contracting into one corner.
(iii) Gas expanding to fill the available volume.
(iv) Burning carbon in oxygen to give carbon dioxide.
Concept: undefined >> undefined
Advertisements
Enthalpy diagram for a particular reaction is given in figure. Is it possible to decide spontaneity of a reaction from given diagram. Explain.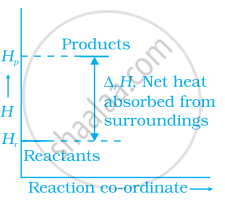
Concept: undefined >> undefined
Match the following processes with entropy change:
| Reaction | Entropy change |
| (i) A liquid vapourises | (a) ∆S = 0 |
| (ii) Reaction is non-spontaneous at all temperatures and ∆H is positive |
(b) ∆S = positive |
| (iii) Reversible expansion of an ideal gas | (c) ∆S = negative |
Concept: undefined >> undefined
Assertion (A): A liquid crystallises into a solid and is accompanied by decrease in entropy.
Reason (R): In crystals, molecules organise in an ordered manner.
Concept: undefined >> undefined
Four structures are given in options (i) to (iv). Examine them and select the aromatic structures.
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
Concept: undefined >> undefined
Write the electronic configurations of the given ions H–.
Concept: undefined >> undefined
Write the electronic configurations of the given ions Na+.
Concept: undefined >> undefined
Write the electronic configurations of the given ions O2–.
Concept: undefined >> undefined
Write the electronic configurations of the given ions F–.
Concept: undefined >> undefined
What is the lowest value of n that allows g orbitals to exist?
Concept: undefined >> undefined
An Atom of an Element Contains 29 Electrons and 35 Neutrons. Deduce
(i) the Number of Protons and
(ii) the Electronic Configuration of the Element.
Concept: undefined >> undefined
Give the number of electrons in the species `"H"_2^+`, H2 and `"O"_2^+`
Concept: undefined >> undefined
A certain particle carries 2.5 × 10-16 C of static electric charge. Calculate the number of electrons present in it.
Concept: undefined >> undefined
How will you convert benzene into p-nitrobromobenzene?
Concept: undefined >> undefined
How would you convert the following compound into benzene?
Ethyne
Concept: undefined >> undefined
How would you convert the following compound into benzene?
Ethene
Concept: undefined >> undefined
How would you convert the following compound into benzene?
Hexane
Concept: undefined >> undefined
Give a brief description of the principle of the following technique taking an example.
Chromatography
Concept: undefined >> undefined
Explain the principle of paper chromatography.
Concept: undefined >> undefined
